Michael Fox
4.9K posts

@foxmdphd
Neurology Prof and Neuroscientist focused on brain imaging and brain stimulation @harvardmed @brighamWomens @brain_circuits





Our Barrow Neurosciences Symposium: “New Frontiers,” is in full swing. Thanks to the over 250 visitors who have come to @BarrowNeuro for this annual event… @foxmdphd








@AndrewZalesky Agree - but this is because we don't think randomized lesions are the best null model. Specificity testing (comparing real lesions causing different symptoms) is a more rigorous control, and was used in all 6 of those studies.







🧠 Breakthrough precision mapping: @foxmdphd and his BD² Discovery Research team at @BWHNeurology are pinpointing which brain circuits drive specific bipolar symptoms—enabling targeted neuromodulation & brain stimulation treatments.


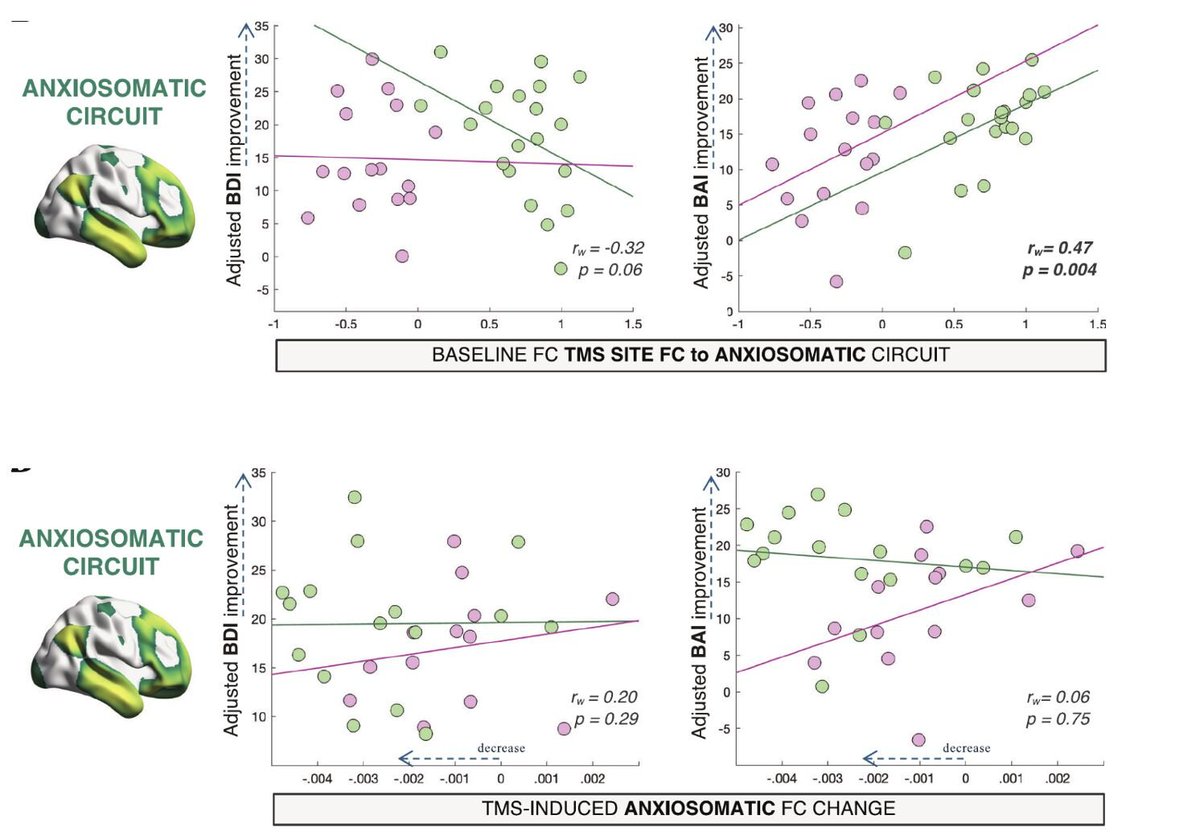

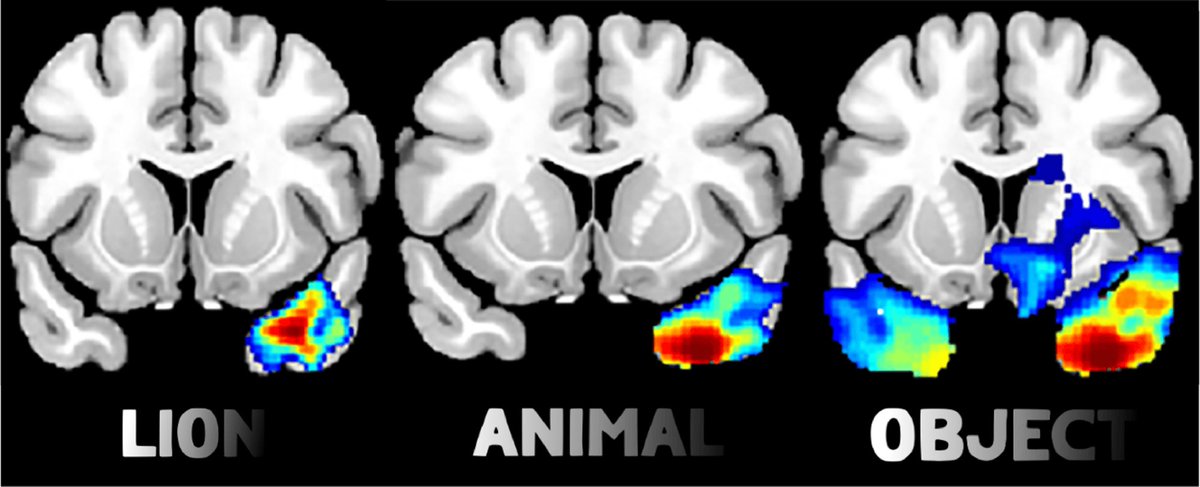

A wave of new papers discuss the merits and limitations of lesion network mapping. By @avaskham thetransmitter.org/brain-imaging/…







Our new viewpoint in @JAMAPsych discussing a new class of biomarkers: “targeting biomarkers”—biological signals that directly guide where, when, and how treatments are delivered. jamanetwork.com/journals/jamap… @foxmdphd @AFornito @Arshiya_San