susan retweetledi

Water is often introduced as something deceptively simple: H₂O, a molecule made of two hydrogen atoms and one oxygen atom.
That description is correct, but it doesn’t tell the whole story. Once you look closer, water becomes far more informative, almost like a record of where it came from.
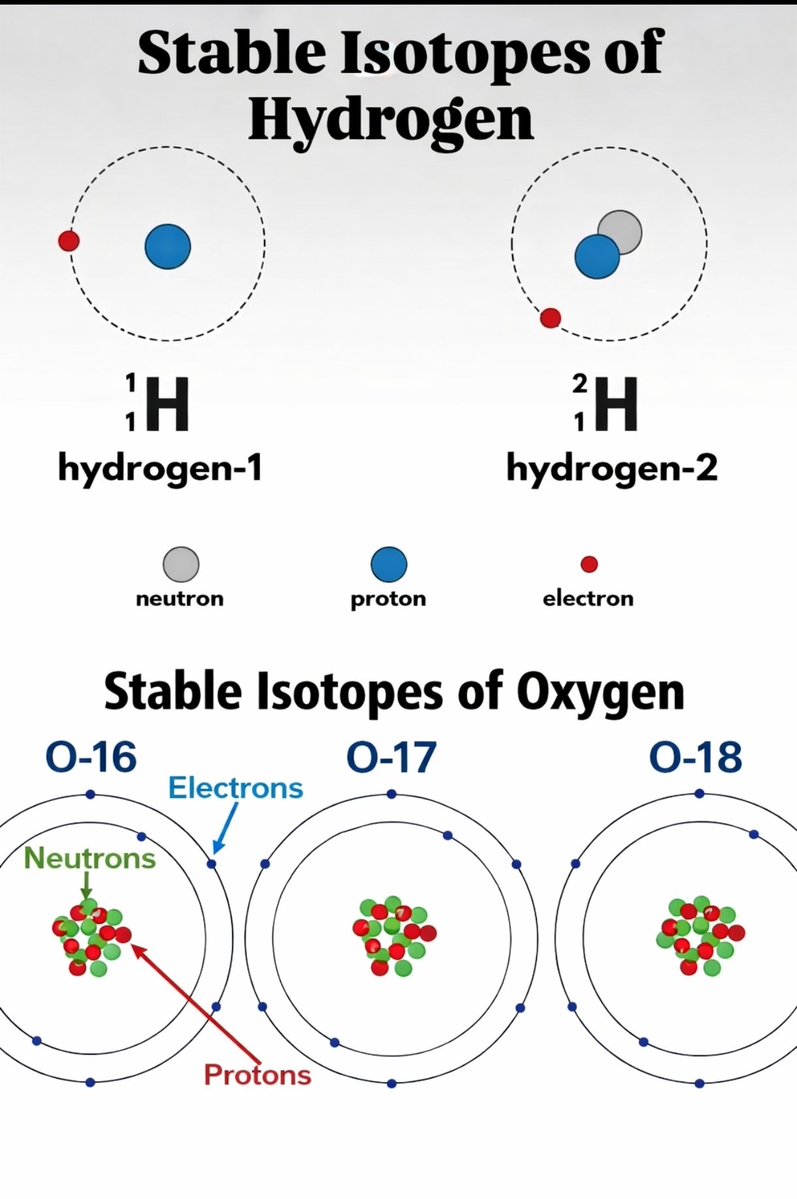
The reason lies in isotopes. Atoms of the same element can exist in slightly different forms, with the same number of protons but different numbers of neutrons.
Hydrogen, for example, can appear as its most common form or as deuterium, which carries an extra neutron. Oxygen also has multiple isotopic variants. When these isotopes combine into water molecules, the result is still H₂O, but with subtle differences in mass and structure.
These differences are small, but measurable, and they give water a distinct chemical fingerprint. One of the most important indicators is the ratio of deuterium to hydrogen. This ratio varies depending on the conditions in which the water formed, especially temperature. In colder, more distant regions of the early Solar System, chemical processes tend to favor the incorporation of deuterium into water molecules, which is why cometary water often shows elevated deuterium levels compared to Earth’s oceans.
Scientists use this fingerprint to trace the origin of Earth’s water. When they compared the isotopic composition of water in comets with that of Earth’s oceans, the match was not as close as expected. Many comets show higher deuterium-to-hydrogen ratios, suggesting they were not the primary source of our planet’s water.
In contrast, a much closer match appears in a class of meteorites known as carbonaceous chondrites. These primitive, water-rich fragments from early asteroids have isotopic signatures that closely resemble Earth’s ocean water.
This strongly suggests that a significant portion of Earth’s water was delivered by asteroid-like bodies billions of years ago, likely during a period of intense impacts known as the Late Heavy Bombardment.
That idea changes how we think about something as ordinary as a glass of water. The water you drink today may not have originated on Earth at all. Some of those molecules likely formed in distant, colder regions of the early Solar System, traveled through space inside ancient rocks, and eventually became part of Earth’s oceans.
So while all water shares the same basic formula, it is not all the same. Its isotopic composition preserves a history that connects it to the earliest moments of the Solar System, turning a familiar substance into a quiet witness of cosmic evolution.
English































