Raffaele Colombo
1.3K posts

@raffcolo
ADCs, targeted therapies, chemistry, biotech, pharma. Associate Director @ Zymeworks



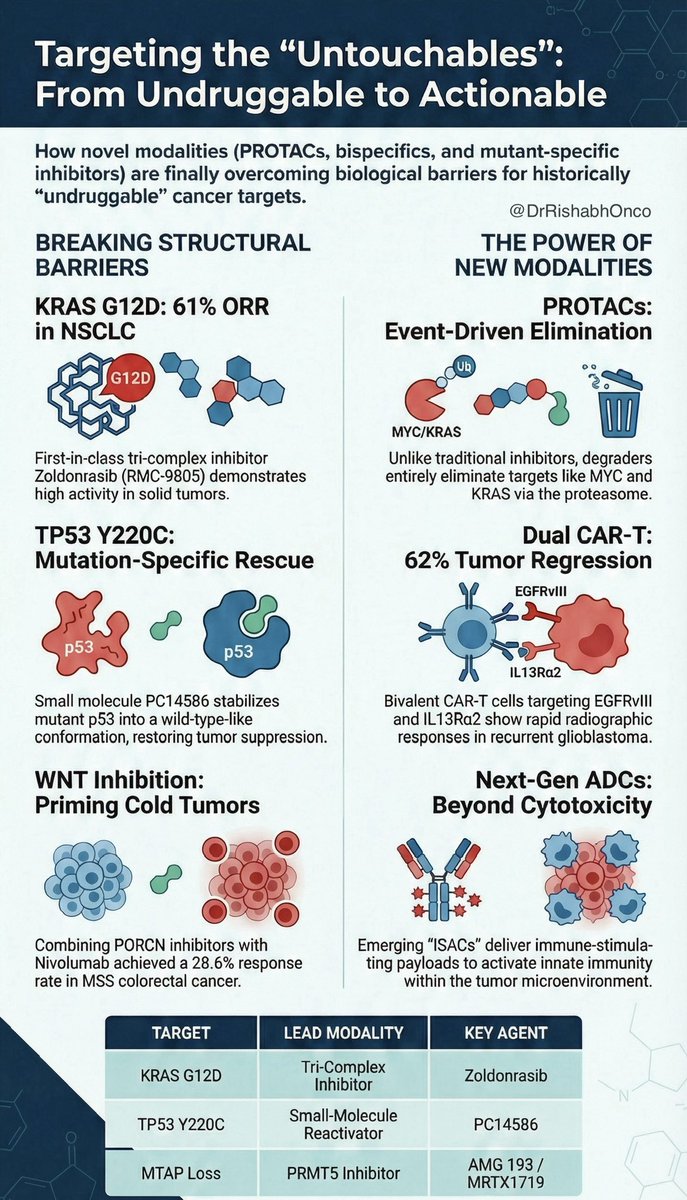
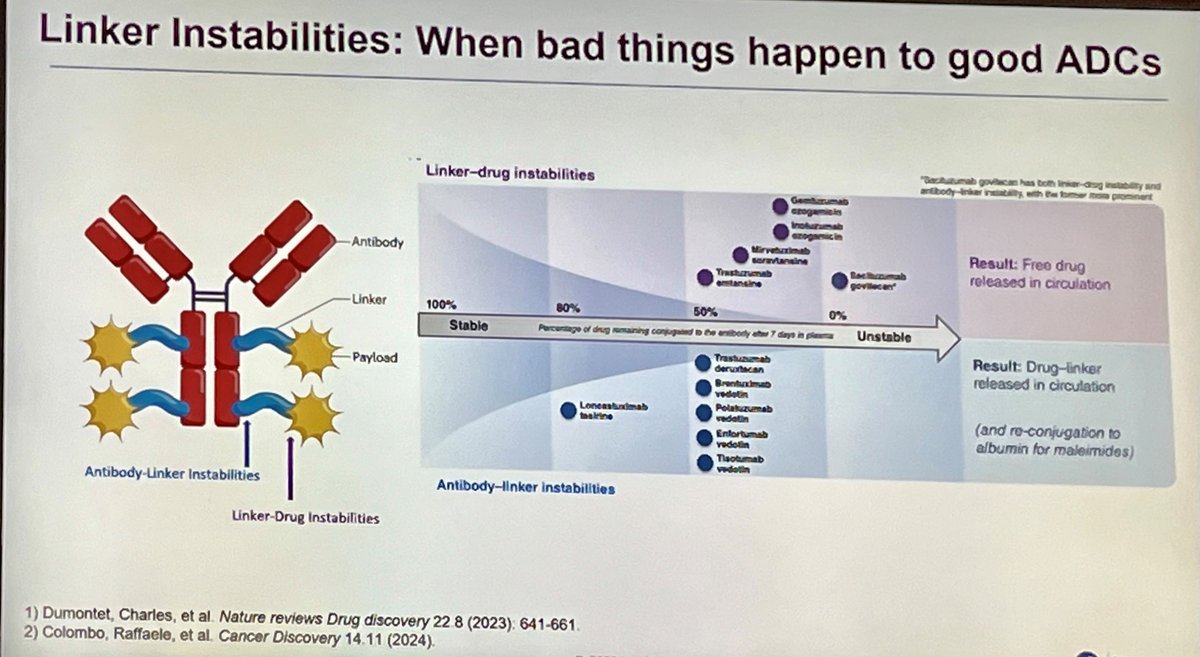
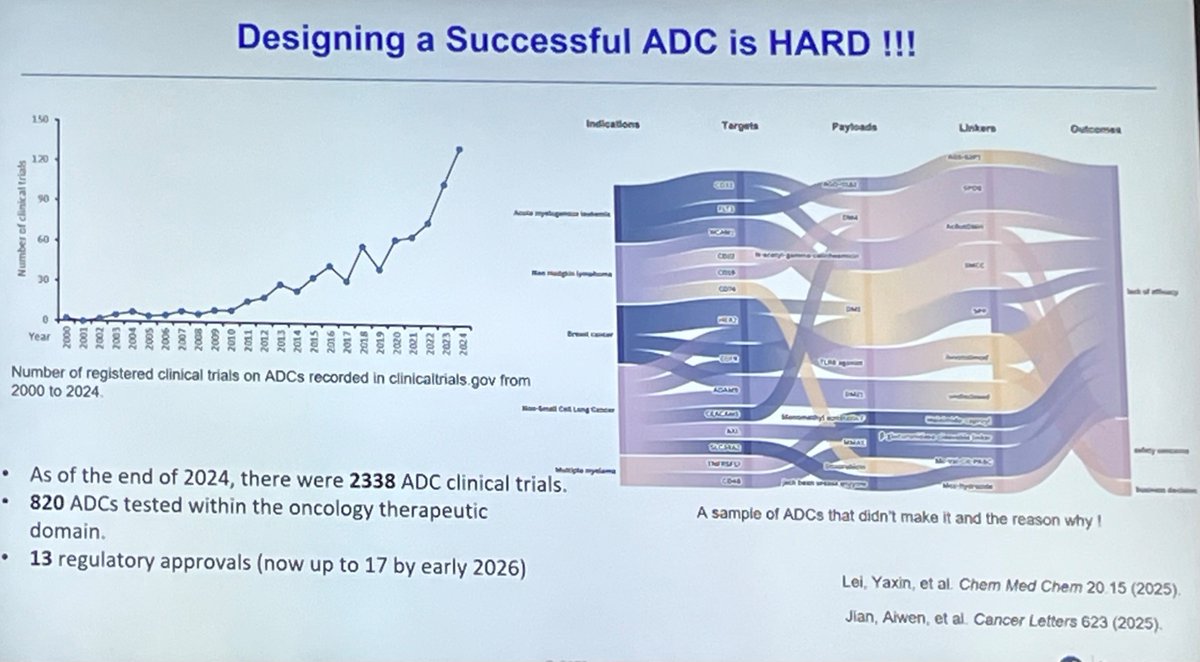
Everyone in the Nectin-4 space is talking about who builds the better ADC. The more important question is who builds the bigger commercial lane. As Padcev's label expands into earlier lines, it creates two distinct populations: patients who haven't seen a Nectin-4 ADC yet, and a growing wave who progressed on MMAE. The next-gen programs chasing Padcev are not equally positioned to serve both. I mapped 130 Nectin-4 programs and isolated payload class as the variable. The landscape splits into two commercial theses. MMAE next-gen programs (CRB-701, 9MW2821, BT8009) are running a tolerability thesis. Better conjugation, cleaner PK, fewer side effects. That is a real differentiator in treatment-naive patients. But it is a single-lane strategy. Site-specific conjugation improves delivery, but does not change the warhead that P-gp efflux and tubulin alterations are already evicting from the tumor cell. The ceiling is displacement of Padcev, not expansion beyond it. Topo I programs are running a different thesis. Because their payload is mechanistically orthogonal to MMAE, they can compete in frontline AND pursue the post-MMAE population. Hengrui's SHR-A2102 showed 39% confirmed responses in prior-ADC patients. Lilly published MMAE-resistant model activity for ETx-22 and LY4052031. These are two-lane assets: first-line competition plus a relapsed population MMAE programs structurally cannot enter. That second lane grows every time Padcev succeeds. Every first-line patient who eventually progresses becomes a patient only an orthogonal payload can serve. Topo I benefits from Padcev's success. MMAE next-gen is constrained by it. The payload is not just a safety variable. It is a TAM variable. The Nectin-4 race is not about who builds the best ADC. It is about who builds the widest one. Comment below for a hi-res PDF of the full landscape analysis.


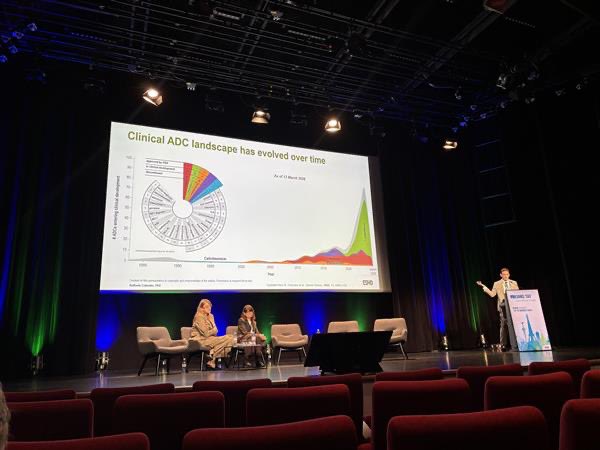











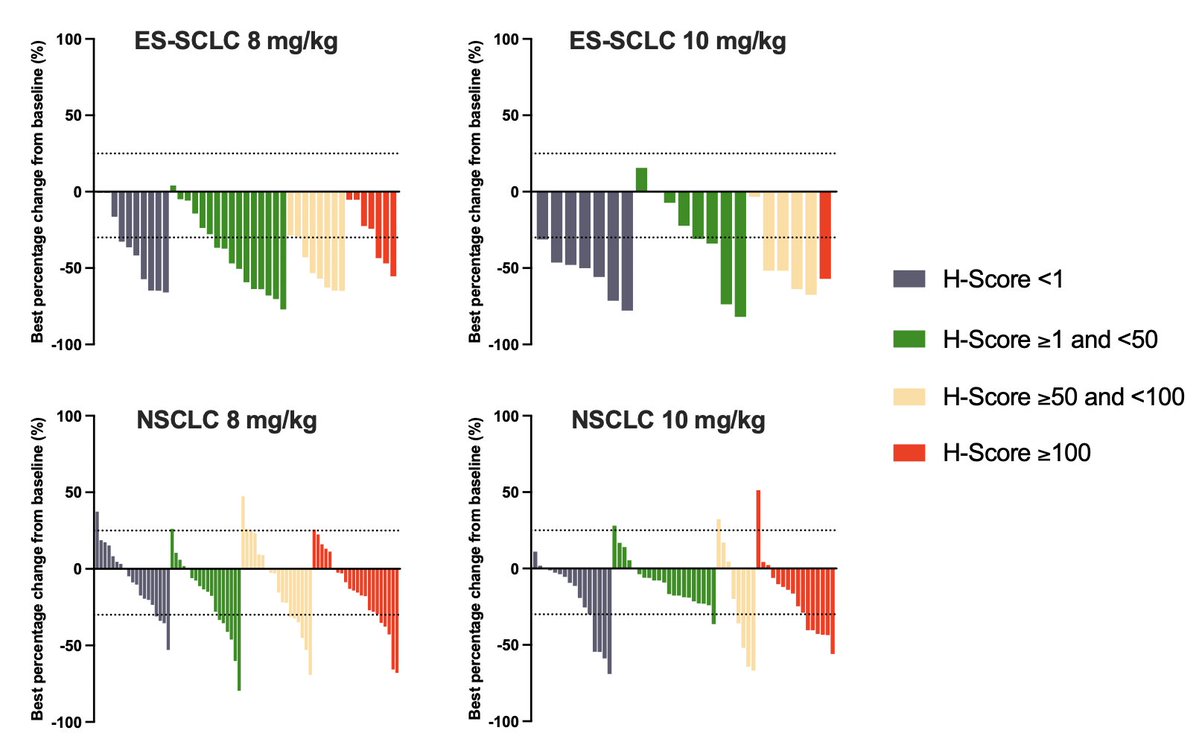
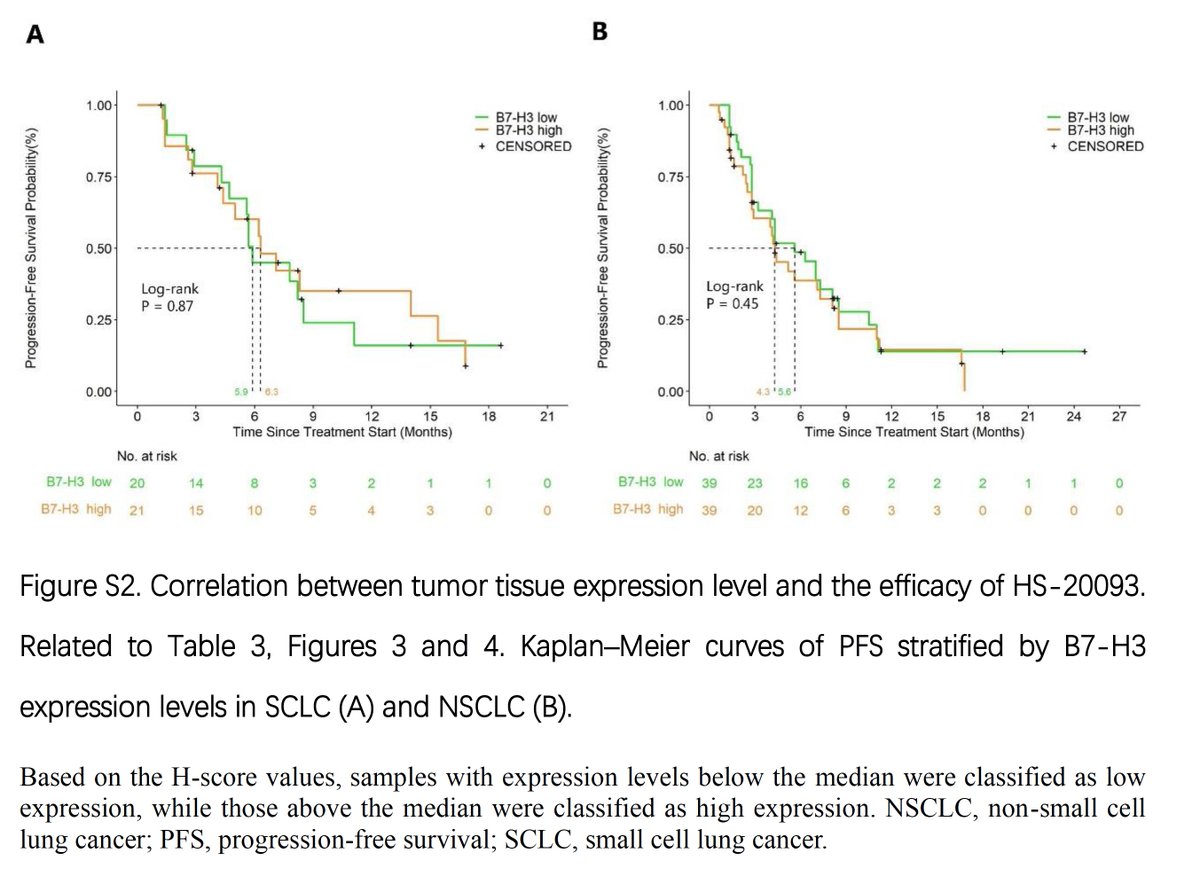
cell.com/cancer-cell/fu… ARTMIS-001, phase 1a/b study of HS-20093 (GSK5764227), a B7H3 TOPO1i ADC, in lung cancer published on @Cancer_Cell 52.3% ORR in ES-SCLC (N=65) 22.4% ORR in NSCLC (N=152) B7-H3 expression levels did not significantly correlate with clinical efficacy







Disitimab Vedotin (HER2/MMAE ADC) was tested in HER2-positive (n=73, RR 55%) and HER2-low (n=78, RR 53%) pretreated advanced bladder cancer outside of China. No clear efficacy difference according to HER status(PFS 6 months and OS 17-20 mnths). Only about 20% of UC patients are HER2 negative. Cumulative neuropathy is like EV, but perhaps less skin rash. It doesn’t have any clear advantages over EV. Sequencing data with EV suggests a lack of complete cross resistant - more date needed). The randomised phase 3 DVP vs. Chemo 1st line trial is complete. #GU26

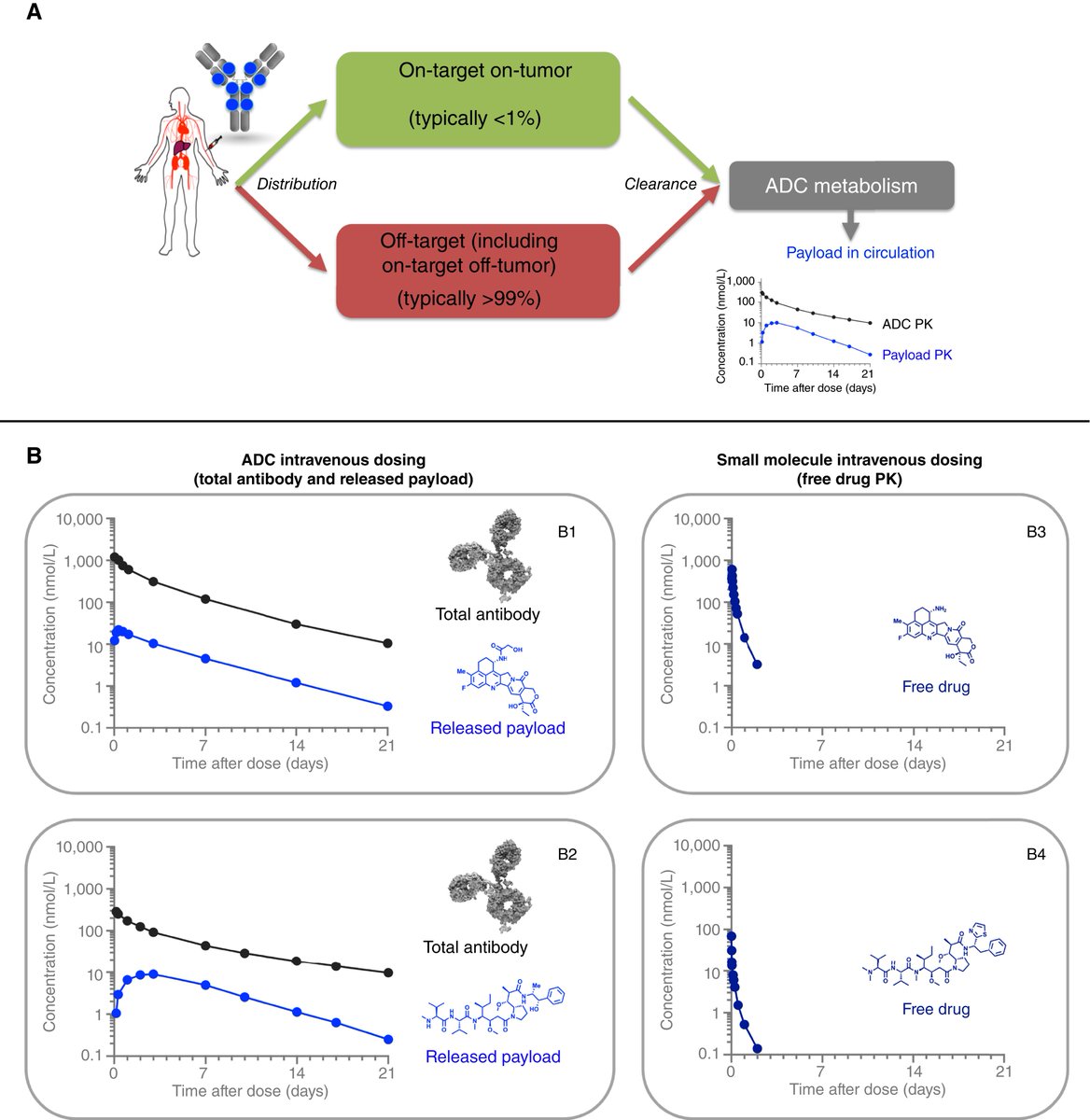
ADC development in China is moving very fast. Global DV monotherapy data #GU26 is hoped to reproduce 🇨🇳 data. In this video Dr Jesse Bain from Shanghai talks about novel topo-1 ADCs & tells us why the linker molecule and metabolism is important. They are not all the same .

This Tuesday 2/24, I am joined by @PTarantinoMD of @DanaFarberNews @DFCI_BreastOnc who discusses "All things ADCs in Cancer." Wide range conversation that may generate more quetsions. Check out this clip, and mark your calendar this Tuesday morning for the full podcast.

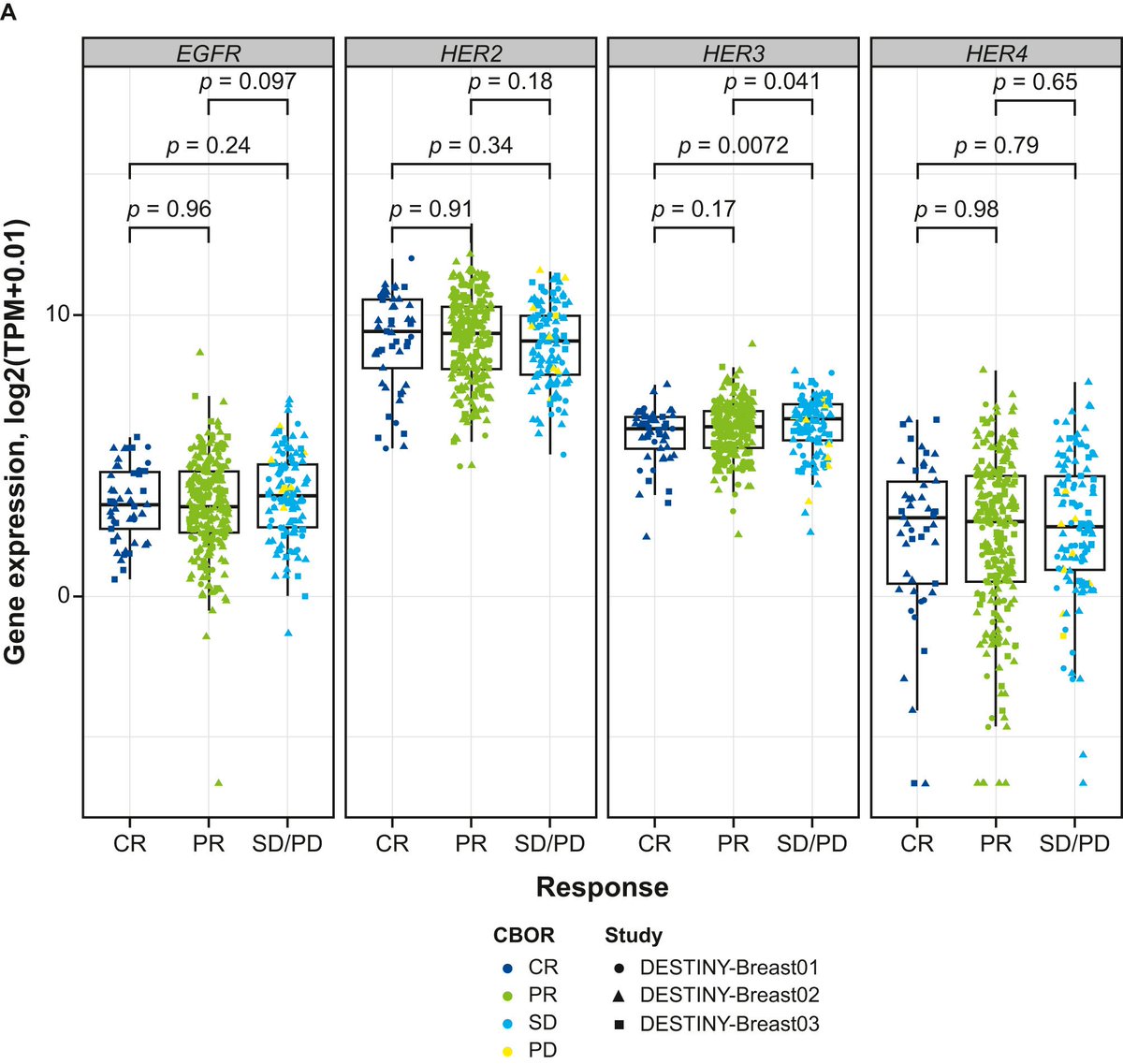
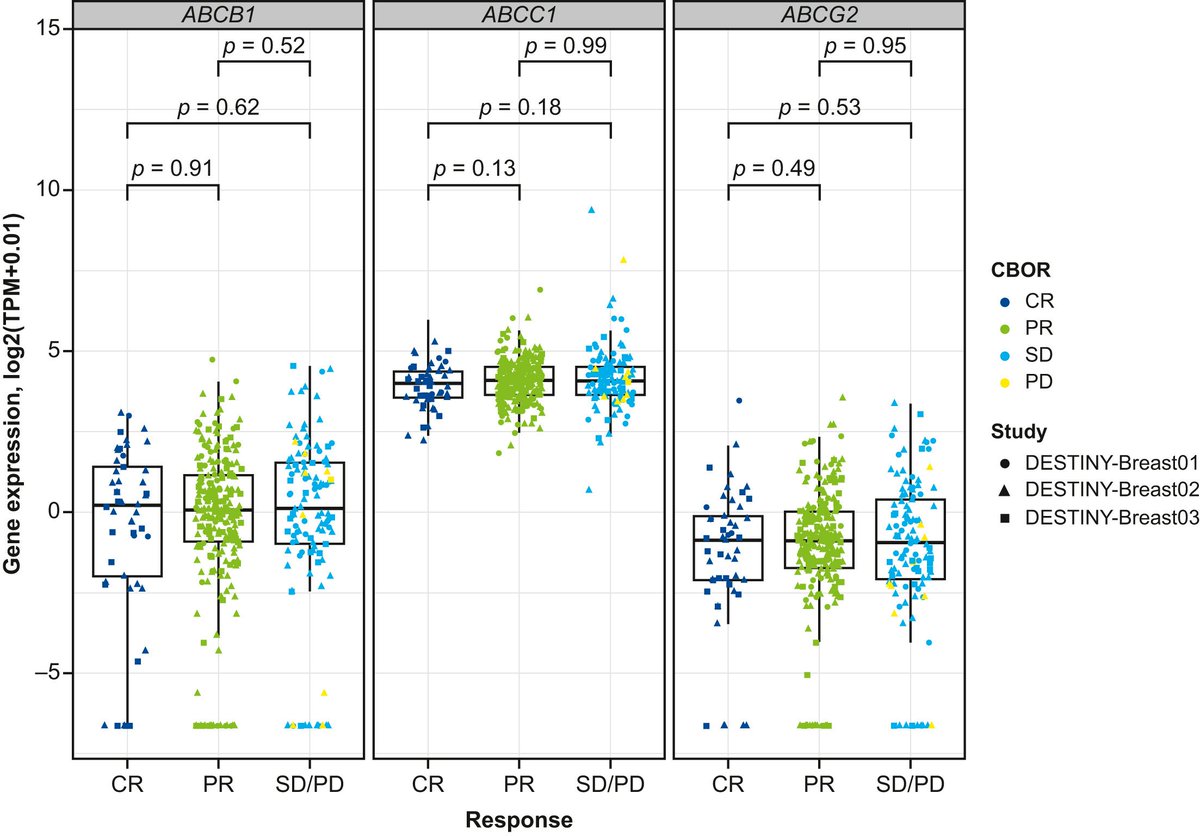
🆕Article of the month podcast🎧: Pooled analysis by best confirmed response to T-DXd and related biomarkers in HER2+ mBC @tompowles1 To read the full article see doi.org/10.1016/j.anno… soundcloud.com/esmo-podcasts/…