
What would be the lifespan of a human who has overcome all aging mechanisms except somatic mutations?
We build a model that estimates it as 140 years, here are details: biorxiv.org/content/10.110…
English
Dmitrii Kriukov
104 posts


@shappiron
Naturephilosopher | Computational Biology of Aging








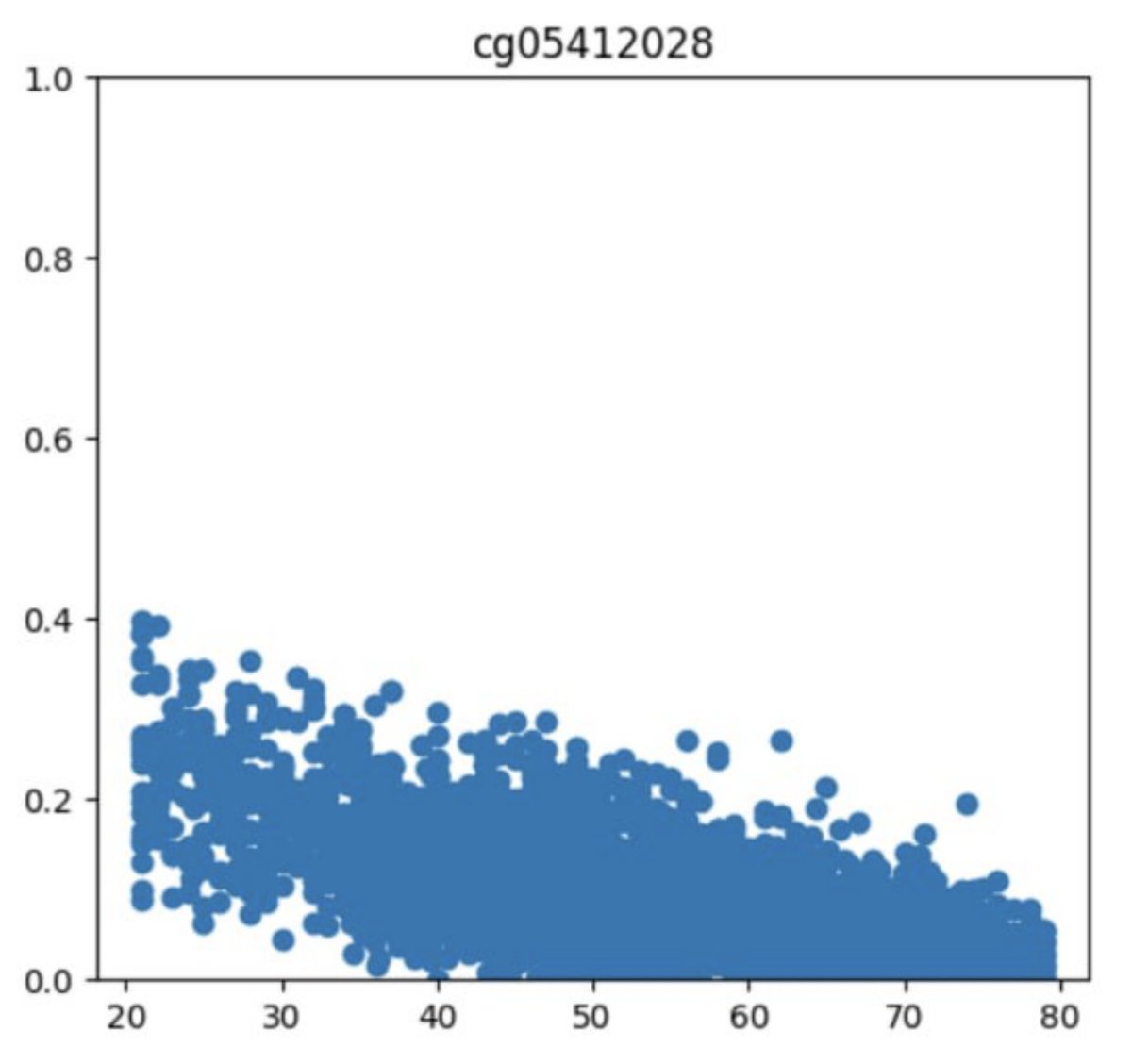












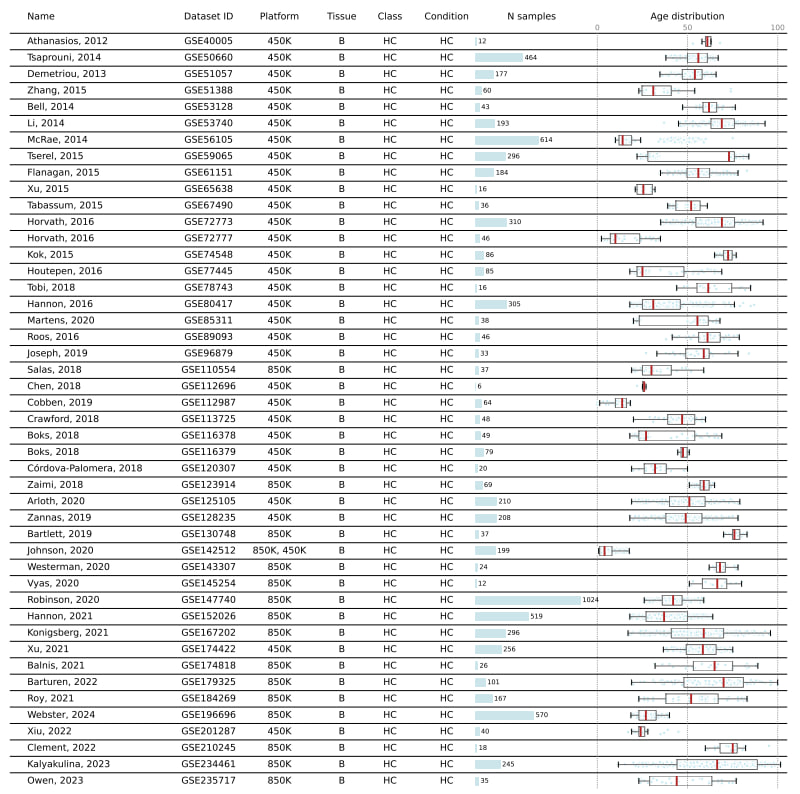

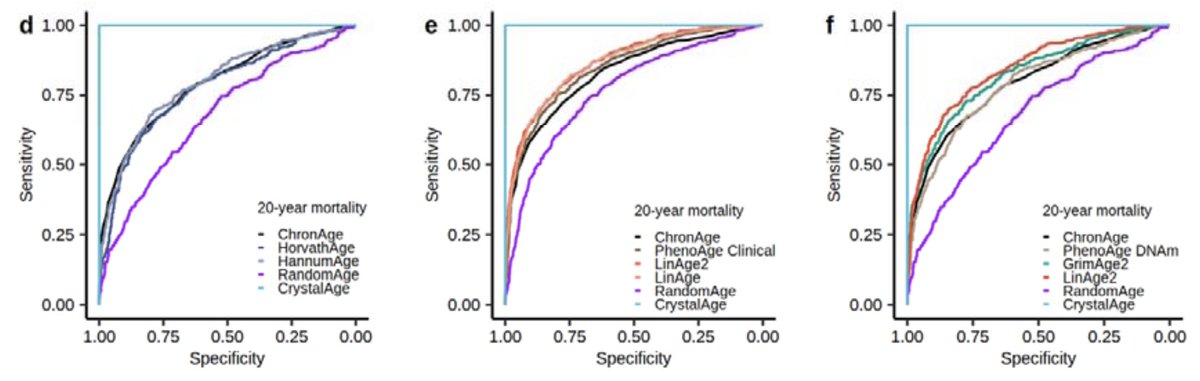
People have been asking us how to interpret linAge, to drop difficult to obtain parameters and to compared to other clocks - we thought it would be a quick job to address these questions - it turned out to be a six month journey - preprint out now: doi.org/10.1101/2024.1…





