Alex Dunn
94 posts

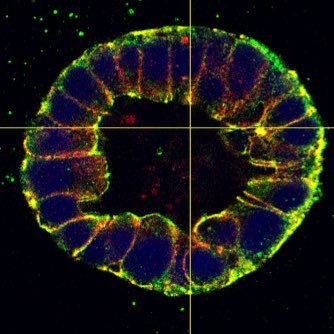
@Dunn_Lab @vvachhar @DejongMatt I read it, fabulous work and really interesting findings, indeed opening many possibilities on how we think about mechanosensing at the junctions. Congrats Alex and team
English

New preprint! @vvachhar and @DejongMatt show that PDZ domains, innocuous-looking scaffolding domains that are enriched at cell-cell junctions, can exhibit striking forms of mechanosensitivity:
biorxiv.org/content/10.110…
(1/2)
English

New paper! @clhueschen presents a mathematical model of flocking on arbitrary 2D surfaces. In addition to your favorite creature, the model can be applied to a broad range of problems in active matter physics, e.g. cytoskeletal flows in living cells.
journals.aps.org/pre/abstract/1…
English
Alex Dunn รีทวีตแล้ว

We micropatterned lymphocytes on EM grids to facilitate cryo-FIB & cryo-ET, revealing networks of filaments on the Jurkat cell surface. Was awesome to work with @dr_slo on this side project during my postdoc in @Dunn_Lab! #teamtomo @AlveoleLab biorxiv.org/content/10.110…

English

@fordycelab Congratulations Polly!!! Great though not unexpected news.
English

New preprint: @bri_zhong shows that pN forces can potentially activate latrophilin 3, an adhesion GPCR that regulates synaptic plasticity. A first step for the lab in understanding this fascinating class of signaling proteins. Congrats Brian!
t.ly/ETL_
English
Alex Dunn รีทวีตแล้ว

#MotorsInQuarantine will feature @amywang01 from @Dunn_Lab and @MarkDodding. Join us tomorrow to discuss how actin links to cell-cell adhesions and kinesin-1 is folded into the lambda particle when not busy transporting cargo along microtubules. mechanochemistry.org/whatson/MiQ/

English

New paper! With the @HTSohLab, we show how single-molecule imaging can be used to to suppress background signal in sandwich-type immunoassays. Congrats and thanks to @AmaniHariri20, @SharonNewman0, and @steventan17 for a great project and collaboration.
nature.com/articles/s4146…
English

Just out: MBoC special issue on mechanobiology! Many thanks to @MattWelchLab and co-editors Dennis Discher, Valerie Weaver, @TheYapLab, and @murrell_lab for involving me in this project.
molbiolcell.org
English

New paper! @bri_zhong describes a new strategy for visualizing mechanical forces across proteins in cells. We hope this will be a useful tool for everyone interested in mechanobiology and are excited to build upon this work in the future.
cell.com/cell-reports-m…
English
Alex Dunn รีทวีตแล้ว

Excited to share that @cavankatenaugh
(@OWeinerLab ), Jennifer Hill (@DavidGDrubin ) and I are organizing the 2022 edition of the Bay Area Cytoskeleton Symposium!

English

Thanks to some good funding news, the lab is looking for postdocs for projects with @LabSkiniotis and Bill Weis (Stanford Struct. Bio.). We'll be exploring ideas I'm quite excited about at the interface of biophysics, cell, and structural biology. Please email for more details!
English

@peiferlabunc @amywang01 Thanks Mark! @ElginKorkmazhan and @vvachhar will be delighted to hear this.
English

New paper! @amywang01 describes the probable mechanism by which alpha-catenin, and likely related proteins, makes a force-sensitive catch bond with F-actin. Also, intriguing clues about the origins of cooperative actin binding. Congrats Amy!
elifesciences.org/articles/80130
English

@jbwallingford Thanks very much! As it happens, was just reading your lab's recent paper on ARVCF just yesterday.
English

Whoa. Cool!
Alex Dunn@Dunn_Lab
New paper! @amywang01 describes the probable mechanism by which alpha-catenin, and likely related proteins, makes a force-sensitive catch bond with F-actin. Also, intriguing clues about the origins of cooperative actin binding. Congrats Amy! elifesciences.org/articles/80130
English

The mathematical tools Cayla and @ElginKorkmazhan developed should be applicable to any situation where noisy single-molecule data are being fit to an underlying statistical distribution. Congrats, Cayla and Elgin! (2/2)
English

New paper! @CaylaMMiller used a clever approach to extract the distribution of actin filament velocities in living cells. The data support a model of the actin cytoskeleton in which periods of mechanical equilibrium are interrupted by sudden jumps. (1/2)
tinyurl.com/4sva4hnk
English

