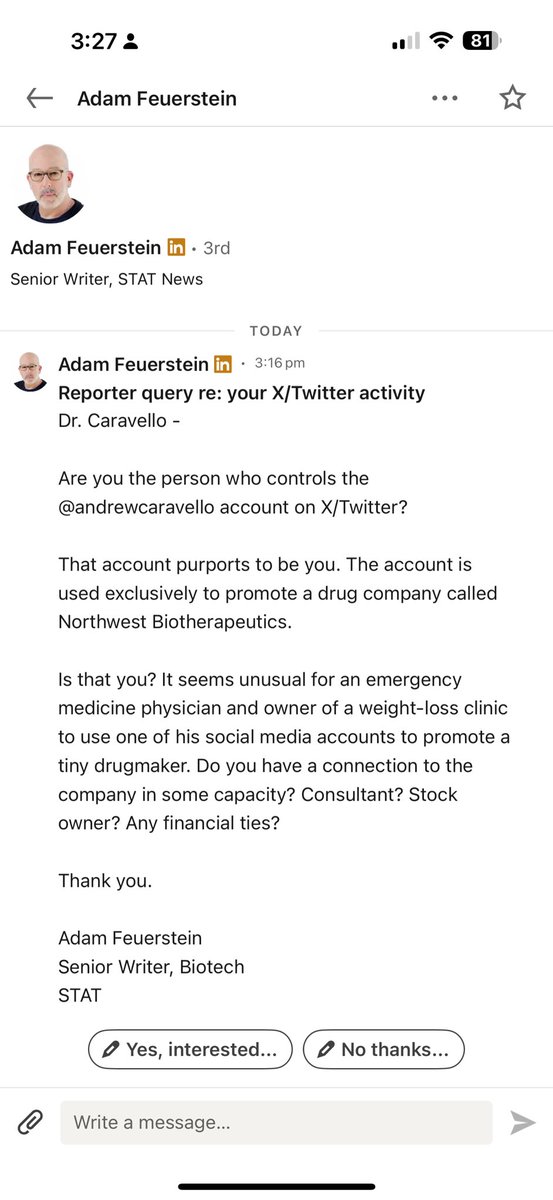
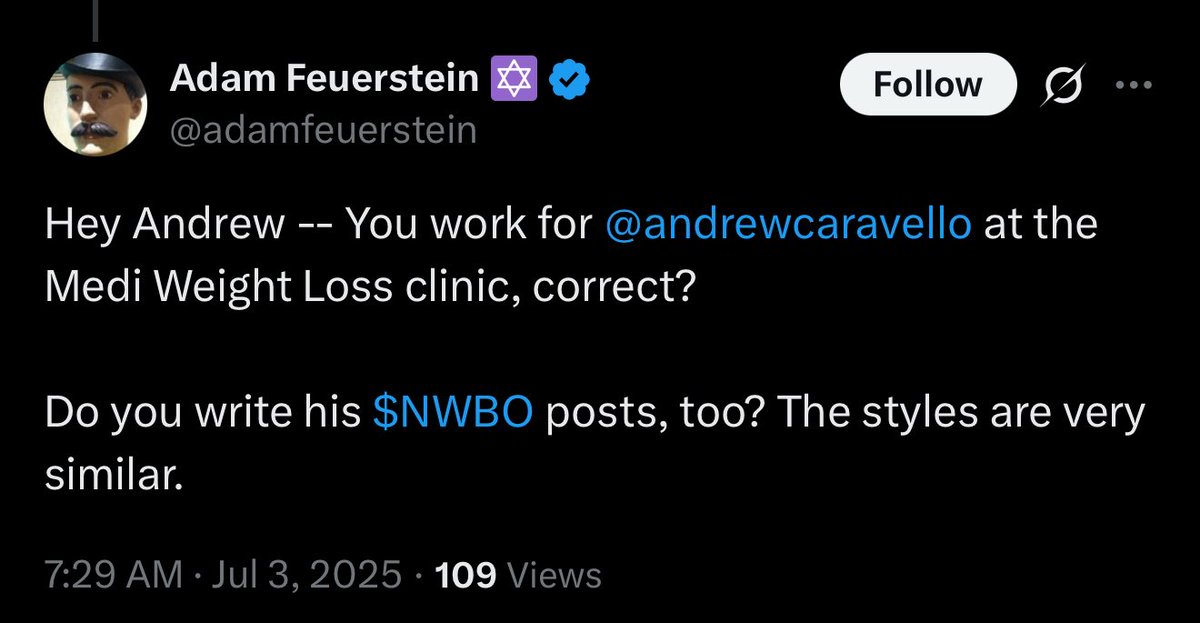
@smith348572 Still the ridiculous spread. Only pre-market and after open we see normal spreads. Sometimes. I had no luck trying to buy shares from 29. Dec - 02 Jan.
English
Spreadburn
1.3K posts


@Spreadburn
Developing trading algos and leading indicators. Mastering quantitative analysis. Swing- and Positiontrader. Focused on Crypto, Tech & Biotech. 9y $NWBO hodler





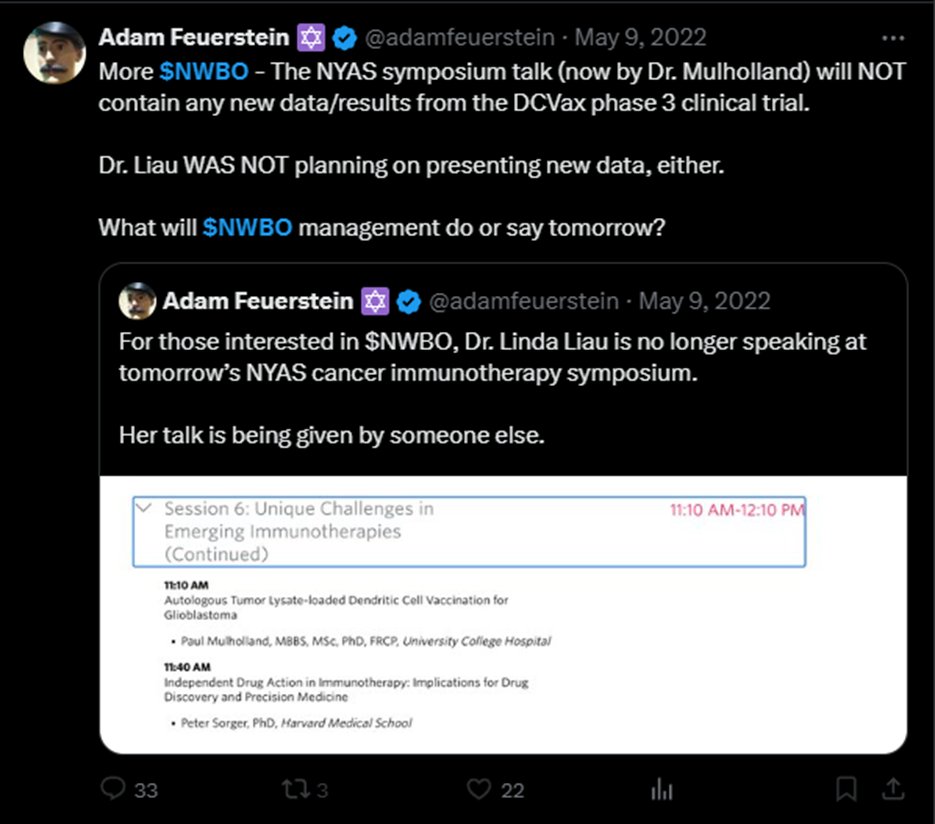


This 👇is why "likes" now mean nothing on X. X is actively suppressing any accounts that talk about stock market manipulation, naked shorting, Virtu, Citadel Reposting is the only way to get around their suppression algo $GNS $DJT $GME $FNGR $GDC $KODK $MMTLP @Christalball93