FlexNP
3.4K posts


FlexNP
@rn_flex
Primary care NP, lover of balanced glucagon agonism, incretin nerd, photographer, mountain lover, coonhound rescuer



$TERN buyout while $VKTX just sits there....













Today, we dosed the first participant in VESPER-5, our second Phase 3 obesity study in just a few months. At least ten pivotal studies are planned for our lead investigational treatment this year, with more than 20 across our ambitious pipeline. We said we'd move fast after acquiring Metsera. We meant it.



Caloric restriction works—but almost no one can sustain it. SGLT-2 inhibitors offer a workaround: force 60–80 grams of glucose into the urine daily, and the body responds as if it's fasting—activating AMPK, suppressing mTOR, upregulating SIRT1, and dampening insulin/IGF-1 signaling. Pharmacological fasting without behavioral change. This week's Research Review examines whether the mechanism is sufficient to extend healthspan. gethealthspan.com/research/artic…






Trying to figure $NVO decision to run several phase 3 on amycretin. That is like $2 B investment ? Probably they feel they should look at everything. In the lancet paper , the escalation was slow. Vomit ( and dysesthesia at higher doses ) were still big problems Look at DC rate among higher doses and even the vomit rate was calculated for all ITT pts ( like 4/16 pts in the 5 mg dose). But they claim a lot of DC not due to side effects ? So the vomit rate is significantly higher than 25% even in the smaller 5 mg. If you look at 20 or 60 mg .. the vomit rate is enormous ( although placebo also had high vomit on this sc trial . but Very small placebo sample ..). Look at slow escalation rate so u can’t improve by further slowing . I do not see it as competitive at all with $LLY tirzepatide Cagrisema combo looks better tolerated and seems best novo bet short term , better that the oral sema excitement that I believe will fade once people try and compare different drugs .. Cagrisema showed high vomit rate in phase 1 but was faster escalation and improved in phase 3 ( was still worse than Sema) So these statements by novo management on amylin improving tolerability are at least misleading. Same going on with $PFE now ).


Amycretin P3 in knee Osteoarthritis Online at CT. Also called AMAZE-5 trial. We have earlier seen some details about this upcoming P3. But there’s more here. Small trial with just 400 patients and 80w. That’s 4weeks shorter than the other P3 trials already up. Could point towards a bit smaller top dose in this trial. 20mg vs 40mg? 3 active dose groups. I wonder if $NVO has started hiding more informations at CT listings. No mentioning about BMI threshold and that participants must want to lose above 25% to apply. Those inclusion criteria were leaked earlier as seen in below post. From NVO investor presentation AMAZE 5 have earlier been listed as H2H with Tirzepatide. But does not look like it from CT listings. I know they will not repeat their mistake from Redefine 4 where it ended as an open label trial since CagriSema and Zepbound use two very different devices. $LLY $NVO $VKTX clinicaltrials.gov/study/NCT07481…



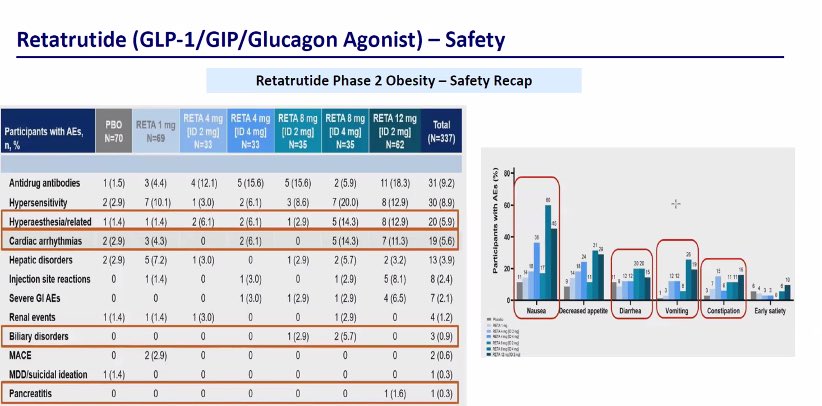
$LLY triple agonist Retatrutide demonstrated significant reductions in A1C and weight in Phase 3 trial for the treatment of type 2 diabetes. $NVO For the primary endpoint retatrutide lowered A1C by an avg of 1.7% to 2% across doses at 40 wks Participants taking retatrutide 12 mg lost an average of 36.6 lbs (16.8%) prnewswire.com/news-releases/…


$LLY In the phase 2 Retatrutide trial 19% cardiac arrhythmias- need to see that safety in this phase 3 trial ???

🚨Big news day: $NVO Wegovy HD (7.2mg injection) has now been approved by the FDA. Novo Nordisk expects to launch Wegovy® HD in a single-dose pen in the US in April 2026. 18.8% weight loss over 72 weeks compared to 15.5% for 2.4mg. (Efficacy estimand was 20.7% for 7.2mg) ml-eu.globenewswire.com/Resource/Downl…