
AdamJButler
1.1K posts


AdamJButler
@AdamJButler
We can make clinical research better for everyone!
Philadelphia, PA, USA Sumali Haziran 2011
282 Sinusundan334 Mga Tagasunod

@RecoveryDoctor These guys are hilarious - won’t even link the original research. “Secondary outcomes generally favored psilocybin over escitalopram, but the analyses of these outcomes lacked correction for multiple comparisons.”
English


@PaulAustin3w This paper clearly shows a negative outcome for psilocybin in this design?? “Secondary outcomes generally favored psilocybin over escitalopram, but the analyses of these outcomes lacked correction for multiple comparisons.” nejm.org/doi/full/10.10…
English

We've been prescribing the same class of antidepressant for 40 years.
Two doses of psilocybin outperformed six weeks of it on nearly every measure that actually matters for living a full life.
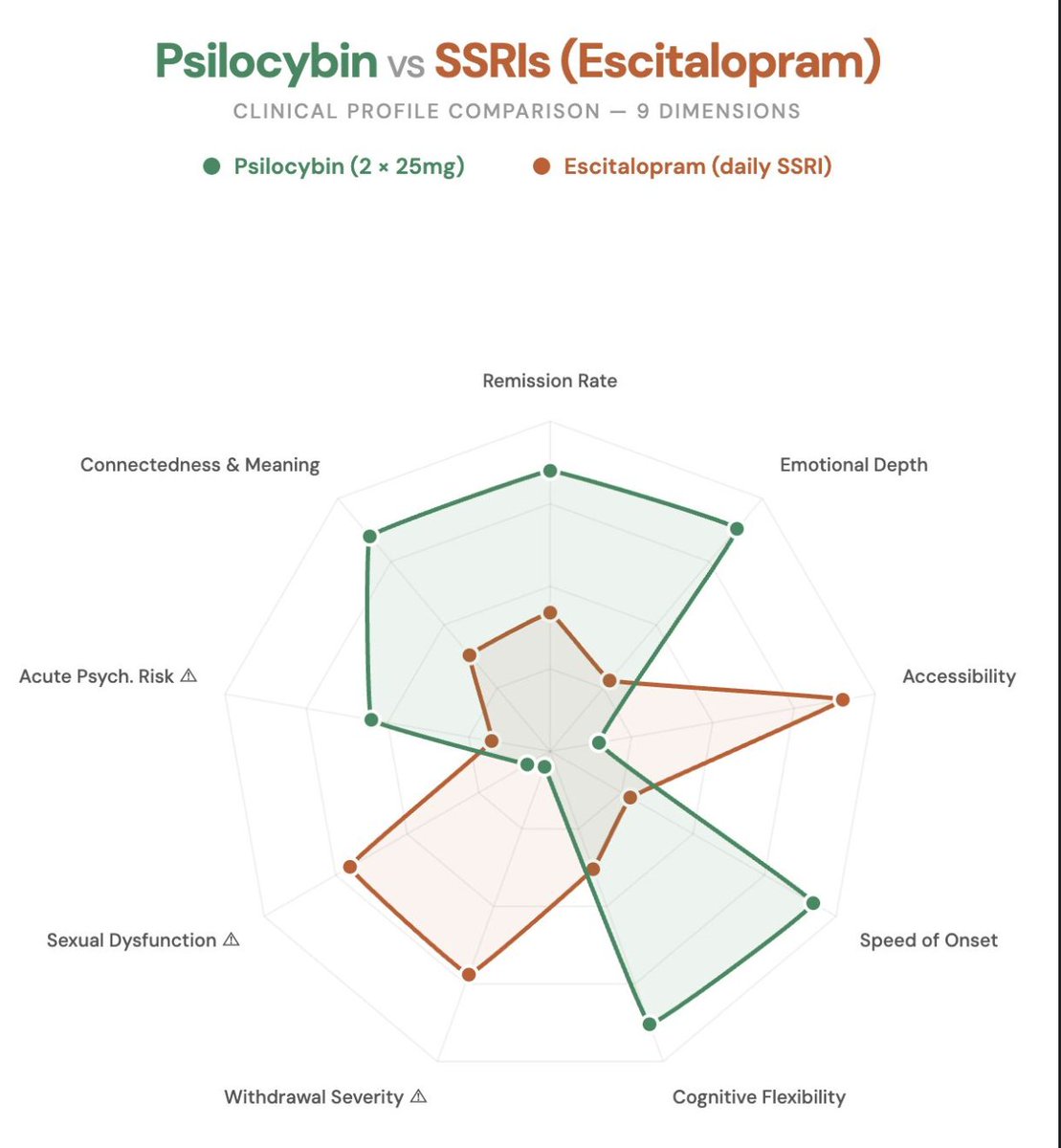
In 2021, Robin Carhart-Harris's lab published what may be the most important depression study of the decade in the New England Journal of Medicine.
A head-to-head comparison of psilocybin therapy vs. escitalopram (a leading SSRI) for major depressive disorder.
Two doses of psilocybin (25mg, 3 weeks apart) vs. 6 weeks of daily escitalopram. Both groups received identical psychological support.
On the primary measure, the scores looked comparable. Headlines called it a tie.
But that's where most people stopped reading.
The secondary outcomes tell a radically different story:
→ 57% remission with psilocybin vs. 28% with escitalopram
→ 70% response rate vs. 48%
→ Psilocybin improved emotional depth; SSRIs blunted it.
→ Psilocybin preserved sexual function, SSRIs impaired it.
Two sessions. No daily pills, no withdrawals, and no sexual dysfunction.
The 6-month follow-up (Erritzoe et al., 2024) confirmed it. Psilocybin showed significantly greater sustained improvements in social functioning, connectedness, and meaning in life.
Not just fewer symptoms but a fundamentally richer experience of being alive (which is what we're all after, if I'm not mistaken).
The neuroimaging explains why. Daws et al. (2022) found psilocybin produced a global increase in brain network integration. The brain became more connected. Escitalopram showed no such change.
SSRIs turn down the volume on suffering. But they don't change the song.
Psilocybin disrupts rigid neural patterns, increases flexibility in brain networks, and as Dölen's research shows, reopens critical learning windows that allow new behavioral patterns to take root.
The SSRI model suppresses symptoms indefinitely. The psilocybin model opens a neuroplastic window, allowing for rewiring and a reintegration of hemispheric balance.
That's symptom management vs. actual transformation.
It's why we built our Practitioner Training at PCI the way we did. If psilocybin opens a neuroplastic window, the practitioner's job is to help clients make the most of it before it closes. Mapping protocols to neuroplasticity timelines while tracking relational & physiological change.
Our next Practitioner Certification cohort kicks off on February 19. I've dropped the link in the comments below if you want to learn more.
What are your thoughts on the future of depression treatment beyond the SSRI paradigm? How quickly do you believe psilocybin will take over as the first line of treatment for the majority of providers?
I'll share my guess in the thread below 👇
English

Publish your data, Chris. “Chen said his company has no plans to publish its data related to GLP-1 outcomes”
Else many will conclude that there is motivated reasoning in full cap risk PCP model behind this.
ChenMed CEO warns against GLP-1s for seniors - blames lean muscle mass loss causing excess falls - endpoints.news/chenmed-ceo-wa…
English

Also a racist who went to the grave believing that Charles Murray’s race science was real. The late Sharon Begley’s obituary tells the story quite well. statnews.com/2025/11/07/jam…
English
AdamJButler nag-retweet

Join academic, industry, and regulatory global leaders at the ISCTM/ECNP Joint Autumn Conference to address challenges and future directions in CNS trials! Amsterdam, Netherlands 9-11 October isctmautumn.societyconference.com/v2/
#ISCTM #ECNP #CNS #trials #methodology

English

@RefiningValue @matthewherper Another data solution built around medical records, claims, EHR. The value proposition for these kinds of data sources is pretty limited and we’ve had hundreds of startups try to show value here in the past 5 years.
English

Laudable goal to improve trial data collection efficiency but I'm not sure how it is diff than existing alts and ultimately faces same ? of whether it's actually replacing any trial collection/cost or just adding on.
But it's Abernathy so worth paying attn to. 🤞something here
Matthew Herper@matthewherper
Amy Abernethy, former No. 2 at FDA, outlines new company’s plans to streamline clinical research statnews.com/2024/10/17/amy…
English

@Sports_bios DELIVER is an interesting use case on a new wearable outcome measure - SV95c. It’s been qualified in EMA as a replacement primary for NSAA in this patient pop. FDA is evaluating it. Good exploratory data in this study could be enough for them to switch in ph3.
English

@ysingh_hopkins Really good paper. There has been similar consolidation of specialty clinics that exclusively serve as clinical trial sites into PE umbrellas. It would be interesting to see if returns have been better or worse than these practices.
English

@erinbrodwin It will be interesting to see if Otsuka can figure out how to combine an app and a small molecule and get the same payor to reimburse for both. The efficacy doesn’t look great compared to existing options.
English

@erinbrodwin @holdspacefree Babylon had peer reviewed data and started as an NHS pilot 🥴
English

Hi we're venture capital and we're here to help with your mental health problems. The wave of VC coming is overwhelming, supported by an entire industry system. VC & "AI" hype selling products point to under-resourced care, seemingly with glee. Related: twitter.com/holdspacefree/…
English
AdamJButler nag-retweet

@matthewherper It begs the question - why isn’t the FDA requiring the use of the CSSR-S in these studies? They utilize this tool broadly (and sometimes punitively) in other indications but I’m not aware of any glp-1 studies that are prospectively measuring this.
English

$LLY’s GLP-1 approved for weight management. Name: Zepbound.
Move over Wegovy? fda.gov/news-events/pr…
English

@matthewherper @23andMe The company is betting the farm on therapeutics. They shut down telehealth earlier this year and personal genome testing revenue is now declining YoY.
English

Interesting window into @23andMe's efforts in drug development: efficacy data on the company's cancer immunity.
52% had stable disease, with median treatment duration of 18.6 weeks. But immunotherapy cohorts can be hard to read.
What do you think?
investors.23andme.com/static-files/1…
English

@Sports_bios One of the key secondaries in this study (SV95c) has been qualified as a primary by the EMA for the same patient population, and is under consideration by FDA. If this separates it could be compelling evidence in support of this application. Patients and families hate the NSAA.
English

This $SRPT Gtx P3 is a reminder how difficult the DMD trials been - .. and also a reminder how STRONG the evidence of the SMA Gtx was - babies w/ type 1 SMA do not survive beyond 13 mos, let alone able to sit independently - .. I am in the camp that for Gtx that works, it really works, it shows, no need to slice and dice.. otherwise.... jmho.
English

@Sports_bios @BiotechObserver I recently rewatched the ALNY adcom. FDA had specific and pointed critique for choice of KCCQ as an endpoint in this patient group. “We are not aware of any evidence of what a clinically meaningful change on the KCCQ is.” All of these other companies have KCCQ as 2nd endpoints.
English

Just think about the FDA cardiorenal division will go thru 3 more reviews/approvals in the ATTR-CM space ( $BBIO $ALNY $AZN/ $IONS) and all of those will be w/ CVOT data - (and label, upon approval will have CV benefit language in there) .. just very difficult for me to see the division to allow non-CVOT data to get thru - UNLESS - the P3 design for $NTLA case is totally different in population - like specifically on tafa failure/intolerant - class III only - .. but the challenges in such a design will be in total uncharted waters w/ risk in clin trial outcomes... so net net - NTLA in ATTR-CM will be a very tough path .. jmho.
English

$NTLA - stk probably will go up if they just forget the whole ATTR-CM space - why bother - .. say the P3 is a win, esp. on the quickie 6MWD type endpt at 12 mos - even allow the FDA for the approval - what will the label be - there won't be any CV benefit data there - while on mkt, potentially w/ Tafa, Acora, Vutrisiran, and the AZN/IONS - can it compete - and IF they need to go thru the CV benefit part - what's the timeline - 4-5 yrs from start of P3 - and roll the dice to on the CVOT odds - .. just a reminder how the population has chged in this space and what kind of clinical efficacy room left for improvement - .. I am skeptical newcomers have much room to wiggle in the NYHA Class I/II population, maybe the Class III side with remaining oppty - but that's an iffy situation w/ +ve clinical outcome... Had $ALNY was able to get thru the FDA w/ the shoddy Apollo-B 12 mos data, there may be a better case for NTLA to muck around over ATTR-CM, now people are rightfully skeptical. jmho.
English

@bijans 13% revenue CAGR and it wasn’t enough - the scale these guys must have been expecting is high!
English

@Farzad_MD It’s a difficult problem. We covered this at @isctm earlier this year - I’m linking the slides. Niacin is commonly used but easy for docs to unblind. These data are pretty robust and don’t indicate placebo response was an issue. isctm.org/public_access/… isctm.org/public_access/…
English

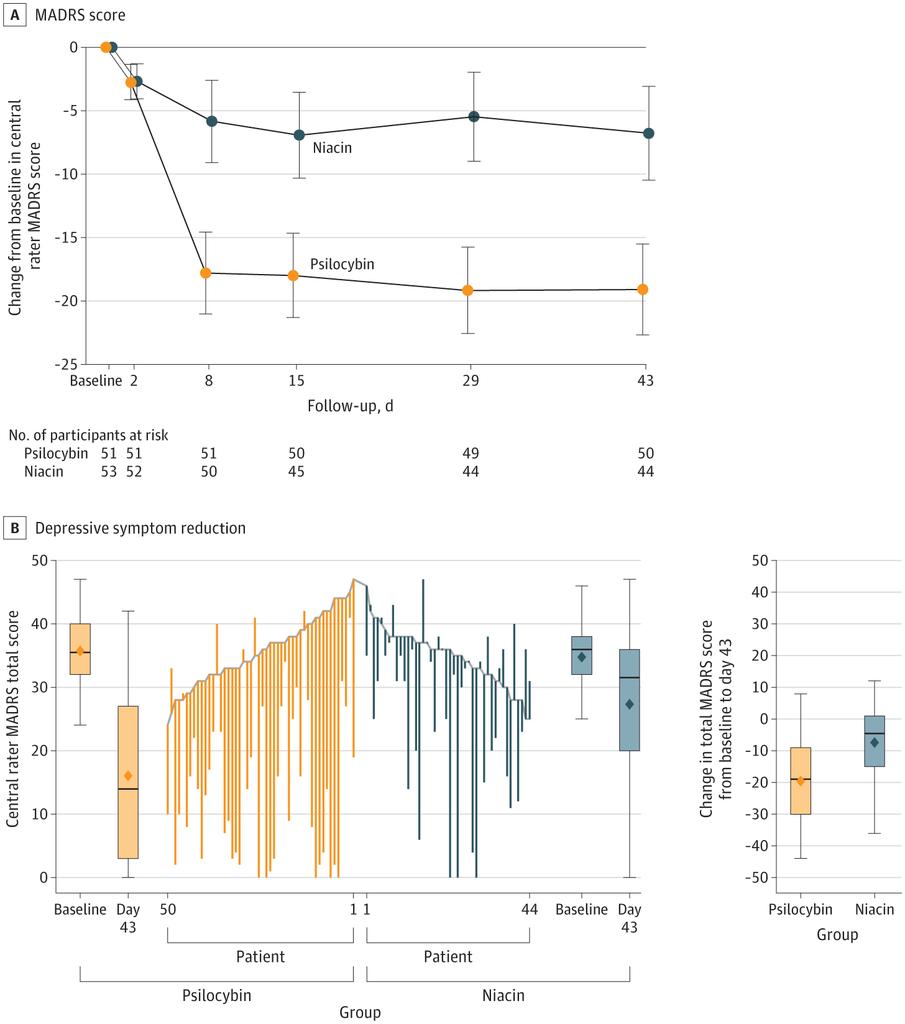
single dose of psilocybin vs niacin (flushing) placebo- powerful results in a randomized clinical trial for depression
how much of this could be placebo effect? (could they stratify cases and controls by whether they "thought" they got active ingredient?) jamanetwork.com/journals/jama/…
English

@erinbrodwin It’s probably notable that these are the data points needed to create a token for a particular patient, similar to what Datavant and others are building.
English




