Alec Plotkin
58 posts

Alec Plotkin
@alecplot
PhD Candidate in Bioinformatics and Computational Biology at UNC-Chapel Hill. Optimal transport, cell fate, and CD8 T cells.
Chapel Hill, NC شامل ہوئے Ağustos 2019
164 فالونگ103 فالوورز
Alec Plotkin ری ٹویٹ کیا

Renaissance of antiviral CD8+ T cell immunity in vaccination and disease dlvr.it/TRZH6K
English
Alec Plotkin ری ٹویٹ کیا
Alec Plotkin ری ٹویٹ کیا

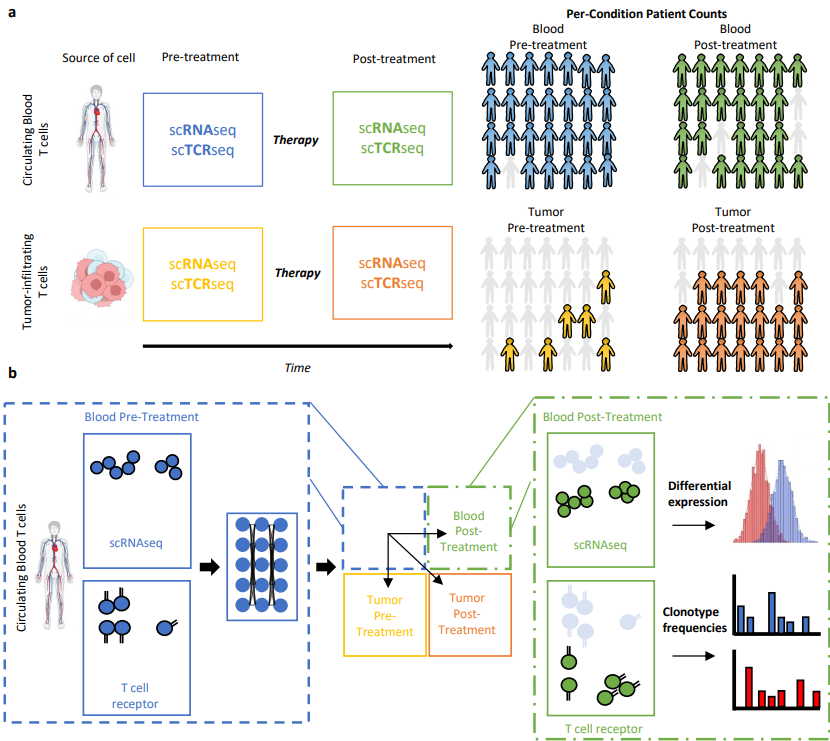
🎉 Congrats to @Schmidt_Center’s @ChujunHe123, @AmodioMatt, Orr Ashenberg, Caroline Uhler and colleagues on their Nature Communications paper! They introduce TRIM, integrating single-cell RNA + TCR data to study immune responses to cancer immunotherapy.
#Abs1" target="_blank" rel="nofollow noopener">nature.com/articles/s4146…
English

Alec Plotkin ری ٹویٹ کیا

Excited to share our new work. Over the past decade, single-cell genomics has transformed our ability to map cellular systems. But a major question remains:
Can we predict how perturbations reshape cellular trajectories over time?
In 2018, we first showed that it is possible to predict cellular responses to perturbations — ranging from disease signals to chemical treatments — even in unseen contexts. In 2022, we introduced CPA (MSB 2022; NeurIPS 2022), extending this idea to predict responses to unseen chemical and genetic perturbations, including their combinations.
Since then, the field of perturbation modeling has grown enormously. The community has pushed the space forward with many creative ideas and powerful models. It’s exciting to see how fast things are moving — even though many fundamental challenges remain.
One of the biggest is that cells are not static. They move through trajectories during development, immune responses, and disease. Yet most current models still predict perturbation effects within a single state, rather than how early perturbations propagate across future states and reshape downstream outcomes.
To address this, we developed PerturbGen, a trajectory-aware generative AI model that predicts how genetic perturbations reshape downstream cellular states.
Huge credit to the people who made this work possible. Thanks to co-first authors @lifeisscience_5, @Adib_m_, @Tomo_Isobe, @Amirhossein Vahidi, @delshadveghari & Anthony Rostron. Special recognition to @lifeisscience_5 and @Adib_m_ for driving this work over the finish line.
Grateful for our outstanding collaborators from @HaniffaLab, @BertieGottgens lab @GosiaTrynka and many others — a true cross-institute effort across @SCICambridge, @OpenTargets ,@sangerinstitute and @Cambridge_Uni.🎉
PerturbGen learns transcriptional dynamics across cellular trajectories. By introducing perturbations at an early source state, it can simulate how these effects propagate into future states along differentiation trajectories.
Scaling this across genes enables the creation of dynamic in silico perturbation atlases — maps of how perturbations reshape biological trajectories over time.
We explored this idea across three biological questions.
First, in a human in vivo LPS immune challenge, PerturbGen predicted that perturbing a transient IL1B signal dampens downstream inflammatory programs in myeloid cells, with pathway changes reversing signatures observed in an independent IL-1β stimulation experiment.
Second, in human hematopoiesis, PerturbGen predicted transcriptional responses to CRISPR transcription factor knockouts and enabled construction of perturbation atlases revealing lineage- and age-specific regulatory programs. These programs could also be linked to human genetics and blood diseases, including recapitulation of signatures associated with ETV6-related thrombocytopenia.
Finally, we asked whether perturbation modeling could help improve complex tissue models.
We built a dynamic perturbation atlas of human skin organoids to identify perturbations that could guideorganoid cells towardhuman fetal skin states.
PerturbGen prioritized activation of Wnt signaling via GSK3β inhibition. Experimental validation confirmed the prediction: treatment with CHIR99021 induced stromal gene programs and shifted organoid fibroblasts toward transcriptional states observed in fetal skin stroma.
Together, these results show how trajectory-aware perturbation modeling can connect gene perturbations to developmental programs, human genetics, disease mechanisms, and experimental interventions.
More broadly, we think these point toward a future where single-cell atlases become predictive systems.
As atlases expand across tissues, developmental windows, and modalities, models like PerturbGen could enable dynamic, virtual perturbation atlases— allowing us to simulate interventions, generate hypotheses, and design experiments before stepping into the lab.
Preprint
shorturl.at/EkisP
Code
github.com/Lotfollahi-lab…
Excited to see how the community builds on this work.
English
Alec Plotkin ری ٹویٹ کیا
New paper: "Distribution-Conditioned Transport"
Modern scientific datasets don't contain one population — they contain thousands. Clones, donors, patients, each its own distribution. DCT learns transport maps that generalize across them, including to distributions never seen during training.
arxiv: arxiv.org/abs/2603.04736
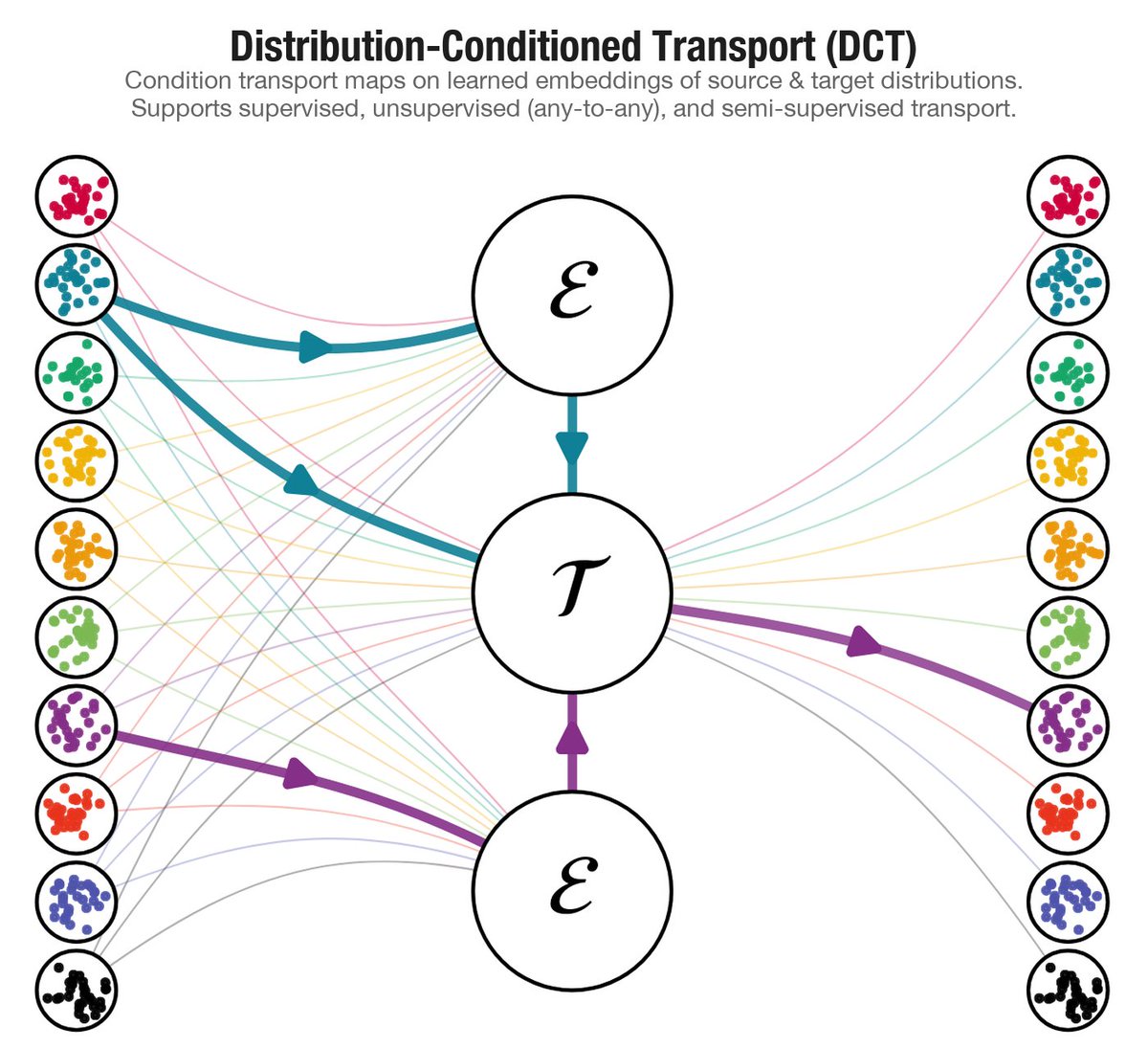
English

@alecplot Not sure, but I would be excited to see this checked!
English

After years of work, the centerpiece of my PhD is published in @NatureMethods! Read it to learn about the biophysical insights we can get from single-cell data!
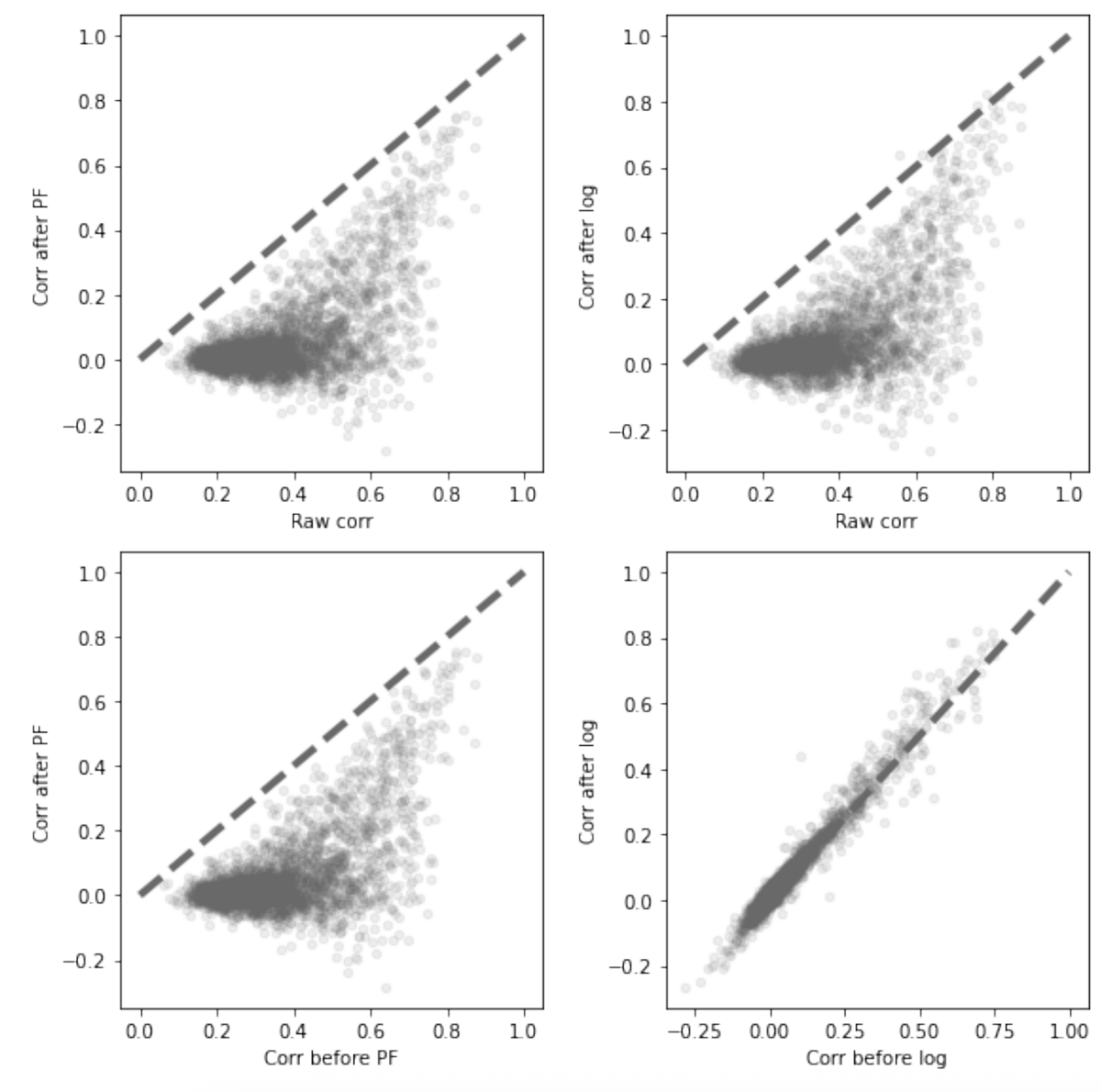
But first, I would like to talk a bit about RNA velocity and normalization. 1/
Nature Methods@naturemethods
Monod fits biophysically motivated models to single-cell transcriptomics data, providing insights into gene expression dynamics. @goringennady @lpachter @mariacarilli @johnjvastola nature.com/articles/s4159…
English
Alec Plotkin ری ٹویٹ کیا

@GorinGennady Do correlations also get scrambled for alternative normalization methods like Pearson residuals?
English

Huge thanks to my my mentors Natalie Stanley (@natstann, @comp_cy) & Justin Milner (@JJMilnerLab) and to my co-authors Genevieve Mullins, Will Green (@greenwilliamd), Huitong Shi, Haidong Yi, and Kay Chung (@HKayChung1) 🙏
10/
English

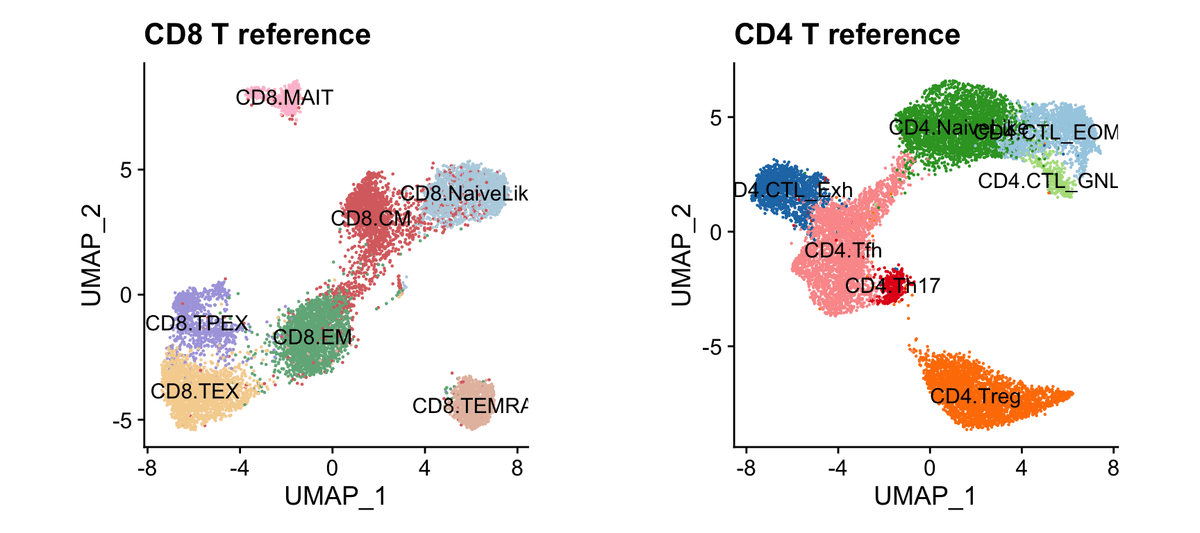
🕰️🚆🦠 Excited to share our new preprint: “Optimal transport fate mapping resolves T cell differentiation dynamics across tissues”!
We use OT to infer realistic CD8 T cell differentiation and migration trajectories from scRNA-seq timecourses.
biorxiv.org/content/10.648…
1/
English
Alec Plotkin ری ٹویٹ کیا

CellPace: A temporal diffusion-forcing framework for simulation, interpolation and forecasting of single-cell dynamics biorxiv.org/content/10.648…
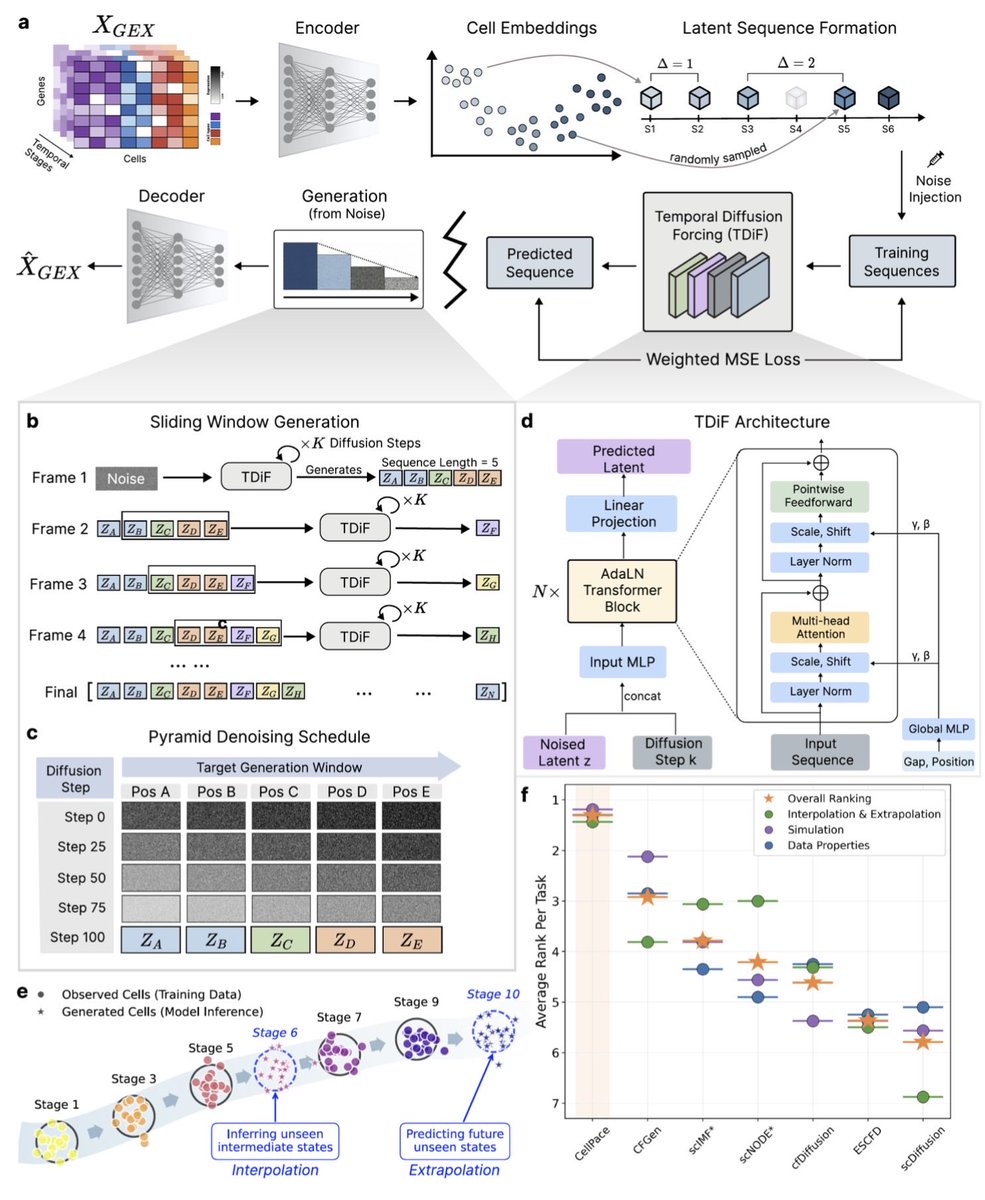
English