autologous assassin 🇺🇸🇨🇱⚜️🏴☠️
871 posts


autologous assassin 🇺🇸🇨🇱⚜️🏴☠️
@DCVaxDefender
$NWBO shareholder. not economic, financial, or legal advice. professional phagocyte. geriatric millennial. semi retired. #whodat




NWBO Spoofing Lawsuit - Joint Status Report $NWBO The Case: Northwest Biotherapeutics, Inc v. Canaccord Genuity LLC, 1:22-cv-10185, (S.D.N.Y.) The Docket: courtlistener.com/docket/6657959… On May 4th the parties in the above action submitted a joint status report advising the Court as to the status of discovery. Both the Defendants and NWBO submitted their own updates within the same letter. The status report contains the typical back and forth bickering. ➡️Defendants state that they are nearly complete with discovery “Defendants’ productions are complete or substantially complete.” ➡️Defendants point the finger at $NWBO for failing to agree on search terms “As of the date of this filing, NWBO has still refused to provide string-by-string hit counts for the terms Defendants proposed in February (even though NWBO confirmed it has run them).” ➡️Defendants object to $NWBO’s 150-day extension request “NWBO had agreed to produce documents responsive to several categories of Defendants’ RFPs in September 2025. Ex. A at 23. Eight months later, it has not done so—and nothing in NWBO’s request for an additional 150 days explains how more time would cure what NWBO has made no effort to accomplish.” ➡️Third-Party Discovery "On May 1, 2026, NWBO filed a letter motion seeking a protective order regarding a subpoena that Citadel Securities served on Joshua Mitts in September 2025." ➡️$NWBO points the finger at Defendants for the discovery delays “The current discovery schedule has been strained by Defendants’ repeated, prolonged, and dilatory discovery disputes.” “The Parties have continued to negotiate and work through numerous discovery issues. But discovery has been mired in disputes largely occasioned or prolonged by Defendants’ conduct.” ➡️$NWBO points at the Defendants’ meager productions to date "Meanwhile, even though NWBO has produced 3,466 documents, no Defendant has produced more than 350 documents. Despite this, Defendants have harassed NWBO with frivolous discovery requests requiring unnecessary burden and expense to contest. NWBO addresses these requests in further detail its motion to extend the case management schedule, which will be filed today." “Moreover, Defendants have limited their data productions to specific days in the relevant period, with some producing only the minutes surrounding the events alleged in the complaint. Additionally, there are inconsistencies between Defendants’ trading data, suggesting curated and incomplete productions.” ➡️My takeaway Citadel, et. al, is attempting to produce minimal documents while running out the discovery clock. $NWBO needs to pick up the pace so they get the documents that are needed. The Court's ruling on $NWBO's 150-day discovery extension along with $NWBO's protective order for Joshua Mitts will be important decisions by the Court.






#dcvax $nwbo #gbm Latest Pawel Kalinski co-authored article:- Type 1-polarized DC immunotherapeutic contains heterogeneous populations with IL-12p70 production restricted to a rare subset: May 02, 2026 "Heterogeneity within αDC1 preparations may underlie inconsistent clinical trial outcomes, and identification of associated surface proteins provides a prospective strategy for subcluster enrichment to enhance DC release criteria and patient stratification for optimized therapeutic efficacy" Gemini AI Analysis: The bioRxiv preprint (DePuyt et al., 2026) titled "Type 1-polarized DC immunotherapeutic contains heterogeneous populations with IL-12p70 production restricted to a rare subset" provides critical insights into the alpha DC1 platform, which is the core technology Northwest Biotherapeutics (NWBO) in-licensed through its deal with Dr. Pawel Kalinski and the University of Pittsburgh/Roswell Park.The paper is highly relevant to NWBO because it identifies why clinical responses to dendritic cell (DC) therapies can be inconsistent and offers a technological "upgrade" to the DCVax platform to ensure higher potency. 1. Synergy with DCVax Platform Technology The DCVax-L platform traditionally uses a standard maturation process for dendritic cells. The synergy here lies in the transition from standard DCs to alpha DC1 (Alpha-type-1 polarized DCs). Potency Enhancement: The paper demonstrates that alpha DC1s are significantly superior to standard "PGE2-DCs" in producing IL-12p70. IL-12p70 is the "holy grail" cytokine for cancer vaccines because it is essential for priming cytotoxic T-lymphocytes (CTLs) that actually kill tumors. Precision Manufacturing: The study uses single-cell multiomics to show that even within alpha DC1 preparations, there is a "rare subset" of cells responsible for most of the IL-12p70 production. This identifies specific surface markers that NWBO can use to enrich the most potent cells during the manufacturing of DCVax, potentially increasing the efficacy of the vaccine. 2. Relevance to the Kalinski/NWBO In-Licensing Deal This research serves as the scientific validation for the "Next-Gen" DCVax platform that NWBO secured in its licensing deal with Dr. Pawel Kalinski.Intellectual Property (IP): The paper explicitly mentions that Dr. Kalinski is an inventor on patents covering this platform and its "upgrade," which have been licensed to Northwest Bio. This confirms that NWBO owns the commercial rights to the specific "know-how" and methods described in the paper to produce high-IL-12 producing DCs. Overcoming "Non-Responders": A major hurdle for NWBO has been explaining why some patients respond to DCVax while others do not. This paper provides the answer: individual variability in the proportion of these "high-producer" DC subsets. By using the markers identified in this study (like Siglec-1/CD169), NWBO can move toward a more standardized, high-potency product. Combination Potential: The alpha DC1 cells described in the paper are shown to be highly responsive to CD40L (a helper signal). This supports NWBO’s strategy of combining DCVax with other agents (like Poly-ICLC or checkpoint inhibitors) to trigger that CD40L response, effectively "turning on" the IL-12 production once the vaccine is injected. In short, the paper provides the molecular "blueprint" for optimizing DCVax-L. It suggests that by selecting for specific sub-populations of dendritic cells identified through single-cell sequencing, NWBO can potentially manufacture a significantly more potent vaccine than the one used in the original Phase III trials. P. Kalinski has been an inventor on other patents covering specific application of available under aCC-BY 4.0 International license. was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made bioRxiv preprint doi: doi.org/10.64898/2026.…; This version posted May 2, 2026. The copyright holder for this preprint (this platform and its upgrade) which have been licensed to Northwest Bio by the University of Pittsburgh and Roswell Park Comprehensive Cancer Center, and he is entitled to portions of the licensing fees and potential royalties collected by both institutions. P. Kalinski is a paid consultant for Northwest Bio. biorxiv.org/content/10.648…


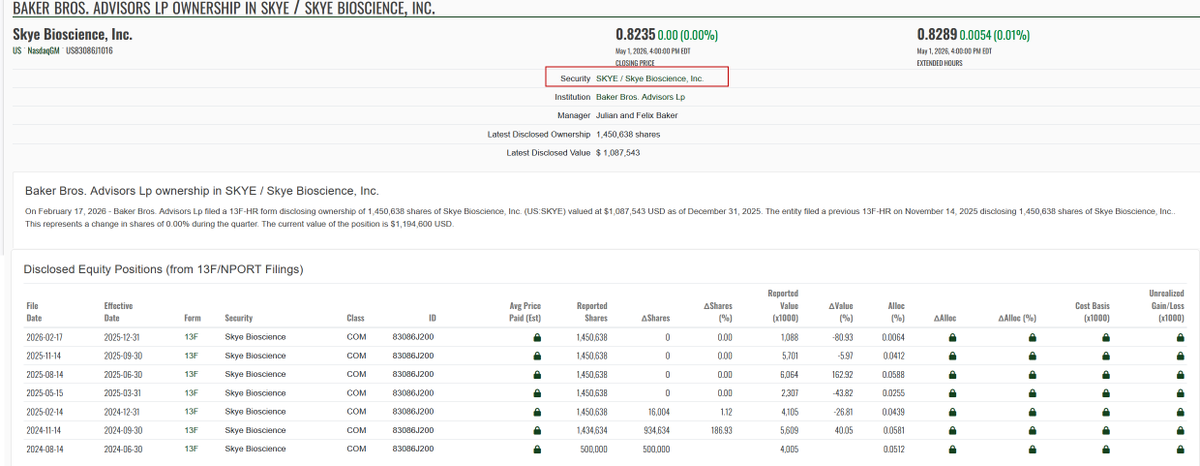
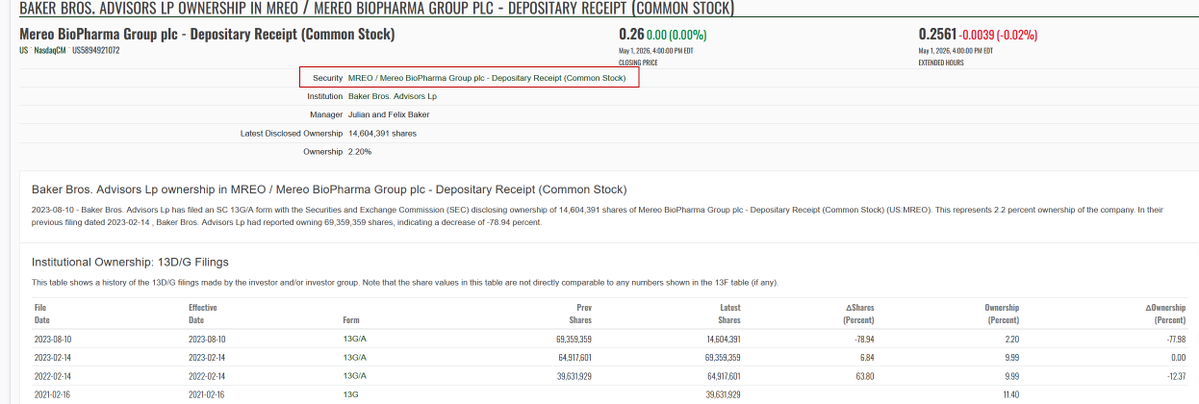
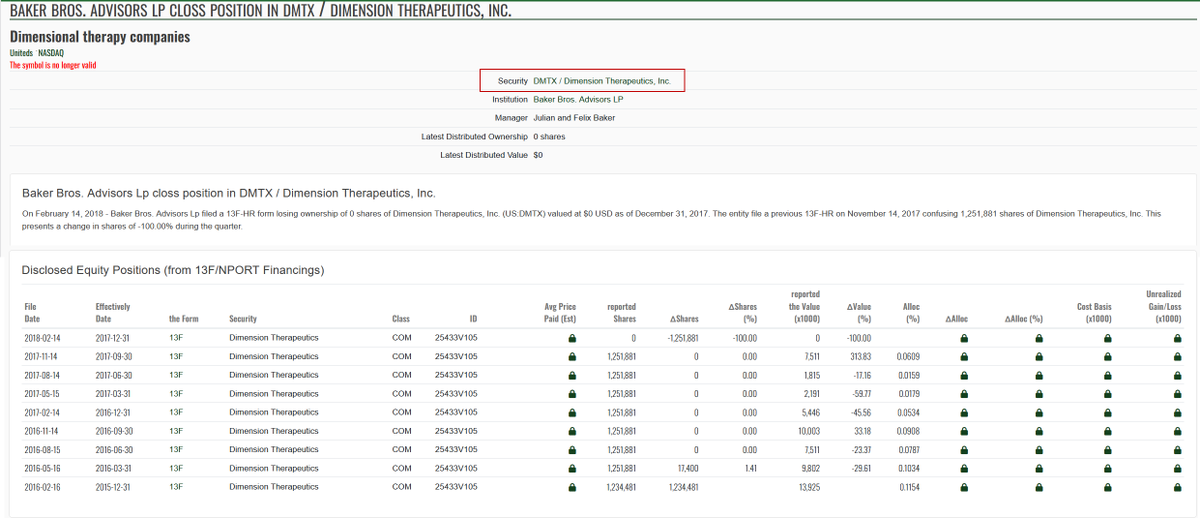

$NWBO Annalisa Jenkins was the CEO of Dimension Therapeutics, Inc and Baker Brothers were the institutional investors. It is a small world indeed!!! Annalisa Jenkins has a stunning resume and it seems like she is going to play a key role in further business operation after the MHRA approval. Hypothetical question: Assuming some big investors agreed on a memo about acquiring a certain percentage of 900m shares, should LP consult with those big investors about recruiting Annalisa Jenkins? Top women in biopharma 2015 fiercebiotech.com/special-report… Northwest Biotherapeutics Appoints Dr. Annalisa Jenkins As Strategic Adviser To Advance Dendritic Cell Cancer Vaccine Platform prnewswire.com/news-releases/… fintel.io/so/us/dmtx/bak…











@hoffmann6383 $nwbo NWBO Just Dimissed the case against one of the defendents "Canaccord Genuity LLC" "NWBO has agreed to dismiss with prejudice all claims asserted against Canaccord in the above-captioned action, without costs to any party" That leaves CITADEL SECURITIES LLC, G1 EXECUTION SERVICES LLC, GTS SECURITIES LLC, INSTINET LLC, LIME TRADING CORP., and VIRTU AMERICAS LLC



$NWBO Annalisa Jenkins was the CEO of Dimension Therapeutics, Inc and Baker Brothers were the institutional investors. It is a small world indeed!!! Annalisa Jenkins has a stunning resume and it seems like she is going to play a key role in further business operation after the MHRA approval. Hypothetical question: Assuming some big investors agreed on a memo about acquiring a certain percentage of 900m shares, should LP consult with those big investors about recruiting Annalisa Jenkins? Top women in biopharma 2015 fiercebiotech.com/special-report… Northwest Biotherapeutics Appoints Dr. Annalisa Jenkins As Strategic Adviser To Advance Dendritic Cell Cancer Vaccine Platform prnewswire.com/news-releases/… fintel.io/so/us/dmtx/bak…