John Cramer, MD
262 posts

John Cramer, MD
@Jdcramer
Assistant Professor at Wayne State University. Head & Neck Oncologic Surgeon. Health services, quality improvement & outcomes researcher. All opinions my own.


💬 Viewpoint: Administrative delays and access failures introduce patient safety risks but are rarely treated as safety events in health systems. ja.ma/4ljWHSu





Can one tonsillectomy approach reduce postop bleeding, shorten recovery, and save money? Dr. Stephen Chorney breaks down coblation intracapsular #tonsillectomy on #ENT259. @UTSWENT @UTSWMedCenter @DrScottFortune Thanks to our sponsor @smithnephew! na2.hubs.ly/H03r5DQ0

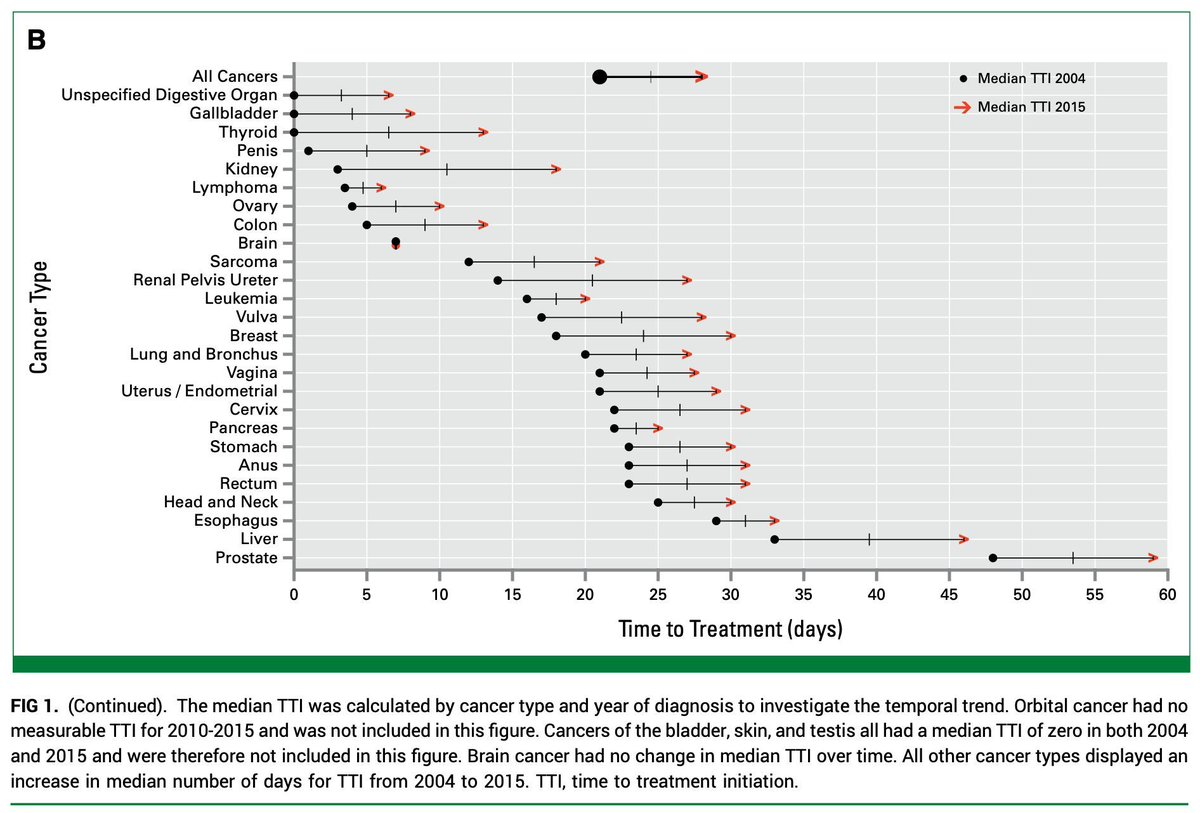



Question for those who’ve dug into KEYNOTE-689: If 43 pts had early ‘clinical progression’ (Table S2B), why isn’t there an early drop in EFS with neoadjuvant pembro? How do you reconcile this?


The California Head & Neck Consortium released the first multidisciplinary consensus recommendations on the use of circulating tumor HPV DNA in HPV+ oropharyngeal carcinoma. The panel of 33 experts representing 15 institutions reached strong consensus that a tool like the NavDx® test should be incorporated into routine surveillance. The consensus recommendations also included: Using an option like the NavDx test improves time to recurrence detection vs. conventional surveillance tools, but should supplement these methods, not reduce or replace them. The recommended time for a patient’s first post-treatment test is 3 months. Serial testing should be performed in conjunction with conventional surveillance every 3 months in the first 2 years post-treatment and every 6 months in years 3-5. For patients with a single positive post-treatment test where no clinical or radiographic evidence of disease is present, the best time to repeat the test is in one month. Earlier molecular detection of locoregional or oligometastatic disease should improve outcomes. These recommendations reinforce real-world clinical validation and value of new technologies like the NavDx test in earlier detection of molecular residual disease and recurrence of HPV+ oropharyngeal cancer. Access the full publication here: bit.ly/3MrPgeG #ctHPVDNA #HeadAndNeckCancer #Oncology #MRD #NavDx #CancerSurveillance #hncancer