Metal
244 posts



@BIOTECHSCANNER @DickieV Whoa! This from StockTwits. stocktwits.com/sixmilliondoll…
and
stocktwits.com/sixmilliondoll…
English

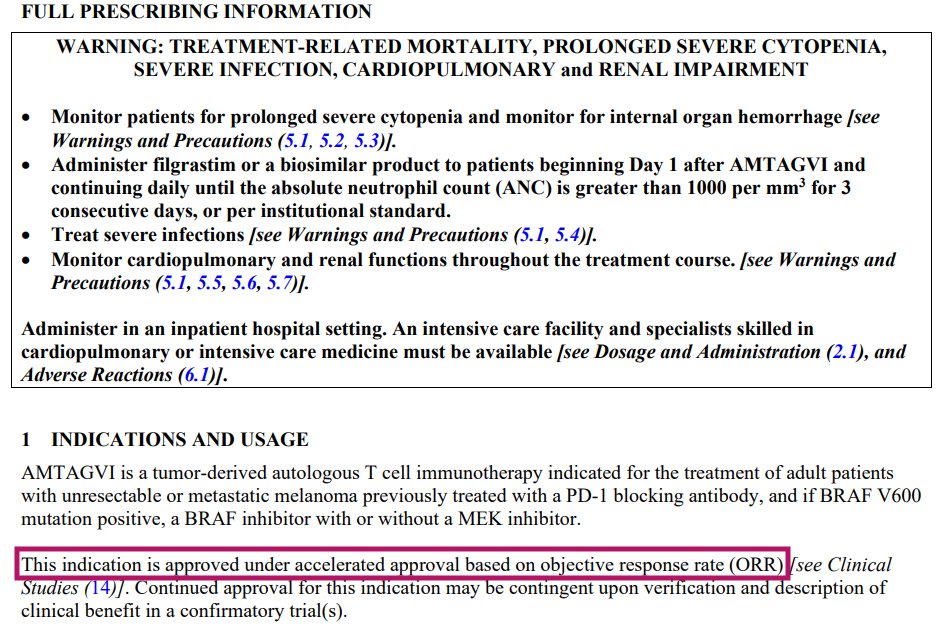
$IOVA - Older patients can still receive Amtagvi treatment with adjustment to lymphodepletion chemotherapy regimen. I hope @DickieV can receive this TIL treatment. It’s a once and done procedure. Other immunotherapy may work but eventually the melanoma comes back. I am praying for this legend to be cured from melanoma with the help of Amtagvi. @IovanceBio hopefully has reached out to him. Amtagvi is awesome baby!
Targeted Oncology@TargetedOnc
Age shouldn't dictate access to cutting-edge care. 🧬 Dr Lilit Karapetyan (@MoffittNews) explains how adjusted lymphodepletion allows patients 70+ to safely undergo TIL therapy. #Melanoma #Oncology #TILTherapy hubs.li/Q04bWv030
English

@Dancingtapas @BIOTECHSCANNER @Bailey13148 ASCO for sure! Saw on StockTwits that late breaking abstracts for AACR are Friday? So maybe?
English

@MetalBiotech @BIOTECHSCANNER @Bailey13148 I don't have that expectation but that would be great place to drop Til 4001
English

@BIOTECHSCANNER
It seems they have 87 ATC"s now per their website. I'm just thinking that maybe that's why management didn't give guidance on the last ER, wanted to get more ATC's up and running before giving more guidance.
English

@BIOTECHSCANNER @Bailey13148 Any indication that they might be presenting at the AACR 17th-22nd?
English

By the time NSCLC is approved, $IOVA should have at least 120 ATCs. They probably have to accelerate ATC approval to accommodate this much larger population. Management wants to give accurate guidance. Shorts start spreading FUD as the guidance must be low. Let shorts pile in. I love what management is doing. They are laser focused on turning $IOVA profitable as soon as possible. This is exactly what I want. I don’t want fluff.
English

@BIOTECHSCANNER Good point. Also I got the impression they’re looking to move it into a different indication.
English

Dr. Gastman said that IOV-4001 is basically a waiting situation but definitely excited about releasing data when ready. But the thing is, IOV-4001 has zero impact on anything really for another 2 years. It’s still very early. I’m excited to get data as well but even if it’s positive, it’s another 3 years minimum for approval!
English

$IOVA - It’s amazing to me how investors let shorts control the narratives about the company. Here are some:
1. When a clinical trial data is not released when expected even though management gave the reason why, then the data must be bad. 😂. Dr. Brian Gastman said “We are very excited about releasing the data on IOV-4001 but data has to come in phases when dealing with the FDA.” The company is waiting on the trial achieving certain milestone to report. We are talking about cancer trials. These take time especially with TIL therapy when DURATION OF RESPONSE is the most important set of data. The more time typically yields better DOR.
2. The company is late on giving guidance so Q1 numbers are probably terrible just like Q1 of 2025. This is what shorts want you to believe. This is after the company showed 3 consecutive double digit quarterly growth! Management even said we are expecting this to continue!!! What the hell else do you want? Oh by the way, management said they are waiting to give 2026 guidance to be more accurate!!! Do you want the repeat of what happened with the previous CFO who gave OUTRAGEOUS UPBEAT GUIDANCE only to get his ass fired and the new CFO had to reduce the guidance by 42% and the stocked tanked by 80%? Is this what you want?
Management is doing the right things for the company and shareholders. I don’t want premature clinical trial data nor inflated guidance! I want real data and real guidance. This is what IOVA management wants.
I can tell you right now that I have heard from the right people that IOV-4001 is saving lives. The data will come whenever it comes.
Ultimately I believe there are too many day traders in the stock. They complain too often and make matters worse. It helps shorts to feel confident actually.
The company doesn’t care about day traders. It’s the long term shareholders that the company wants to serve. As CEO Fred Vogt said, “Iovance is poised to create substantial value for patients and shareholders as we increase revenue…”
3. The FDA is tough on REPL, it will be hard on $IOVA. Really? Have you seen what type of experts the company has hired recently? Have you bothered to look? They have some of the top experts that were at the FDA for years and they still have great relationship with the organization. Dr. Brian Gastman said, “We are in constant communications with the FDA.”
What else do you want? I know. You want the share price to drop to $3 so you can load then sell at $4. That’s what you want. Let’s be honest😂.
You are not going to find a biotech company with more potential to 10X in the next 5 years than $IOVA.
You won’t. There are two leading edge adoptive cell therapy platforms right now: CART-T and TIL. TIL is for solid tumors that make up 90% of all cancers. Guess what company is THE ONLY ONE WITH AN APPROVED TIL TREATMENT? Yes, $IOVA.
If you are an investor, stop complaining. Maybe send a message to @IovanceBio and thank them for what they’ve been doing for patients and shareholders.
Be patient. Focus on the long term goals of the company. This is the one.
GIF
English

@AlpBugraBasat You should probably check the dates. I was in IBIO under .75 & sold at $3 😂 Now learn how to read a balance sheet!
English

$REPL I’m now long. The pendulum has swung too far. RP1 monotherapy showed 35% ORR, with a majority being CRs, so this isn’t a Nektar-style combination hype.
CDER will also soon have new leadership with deep, hands on experience in cell and advanced therapies.
Lastly, $REPL has $300M in cash versus a $140M market cap.
English

@robertcoffin3 Right! We should listen to those “arrogant” letter writing experts instead, all of whom have massive conflict of interest which they didn’t disclose. Bottom line is it wasn’t a good trial. linkedin.com/pulse/replimun…
English
As Philip said on CNBC re the $REPL CRL, if the FDA have concerns about the clinical meaningfulness of Replimune's data, the correct way to sort that out is to hold a traditional advisory committee meeting where actual experts can opine - not the FDA arrogantly determine that they know better than everyone else, including having ignored the multitude of letters written to the FDA by a who's who of melanoma experts, all of whom know the data in detail, after the first CRL.
English

@twotreesthere @US_FDA Ignorant post. IL2 has been given with TILs for decades as a boost. Amtagvi BLA is the regimen of lymphodepletion-TIL-IL2. Further more IOVA ran a good trial, true salvage population/no heterogeneity issues. Due some dd.
English

$REPL If the current @US_FDA wishes to do away with accelerated approval path for unmet medical needs, they should have the courage to stand on their own reasons.
IL2+Amtagvi was approved on single-arm ORR data via accelerated approval in 2024
PR1+Niv rejected for 2021 feedback?



twotreesinthecenter@twotreesthere
For PD-1 failed advanced melanoma patients, $IOVA's TIL+IL-2: ORR 31.5%, CR 4.1%, 7.5% treatment-related death (n=160) $REPL's PR1+Niv: ORR 32.95%, CR 15%, NO treatment-related death (n=141) Thanks to #FDA @VPrasadMDMPH the latter, unavailable.
English

@Bernard22190947 @Henry431171040 @adamfeuerstein What?! Amtagvi is already approved.There is no other component. Heterogeneity wasn’t/ isn’t an issue because it’s a true salvage population. The surgical excision is to harvest TILs. It’s before treatment not during or after. Not a single thing you said is correct
English

@Henry431171040 @adamfeuerstein Yea but $iova drug will run into the same issues; contribution of components, patient heterogeneity, surgical confounding…
English

$REPL FDA rejected RP1
CRL letter
download.open.fda.gov/crl/CRL_BLA125…
English

@RNAiAnalyst @AlexandrG1980 That argument doesn’t work. Those choices were made well before Amtagvi was approved. And how can those medical societies do that when the data is so confounded?
English

@MetalBiotech @AlexandrG1980 Let the medical societies figure out when in the treatment paradigm to use RP-1.
Also, do you think trial investigators would have chosen the combo when they thought something else was indicated?
English

@RNAiAnalyst @AlexandrG1980 I’m not making your point. We don’t know how well RP1 works in the same indication where AMTAGVI works and works much better.
English

@MetalBiotech @AlexandrG1980 You are making my point. RP-1 works...approve it.
English

@RNAiAnalyst @AlexandrG1980 Recent real world data for AMTAGVI ORR 44%. And when used as true 2L ORR rises to 52%. So how does FDA approve RP1 w unclear effect which could take away from something else with clear & better data?
English

@MetalBiotech @AlexandrG1980 Are you saying RP-1 is ineffective and didnt contribute to the >30% ORR in the PD-1 failed population?
English

@RNAiAnalyst @AlexandrG1980 We get it. You have issues with the FDA. BUT look at the trial. It was very flawed. That much is incontrovertible, regardless of FDA communications or lack there of
English

@AlexandrG1980 So the FDA let Replimune interact with the old review team without telling them of the new team? That would be the 🍒 on the cake. $repl 🤣
English

@MaverickNY @robertcoffin3 Robert, you’re making an assumption that Sushil is being forthright. Why is that your knee jerk reaction? Anyone with half a brain can see that this was a very flawed trial!
English

@robertcoffin3 Not making any assumptions about the current FDA, although have not always been a fan. I would have expected more substantive push back on the methodological issues they highlighted though.
English

@avidresearch @MaverickNY The FDA saved lives. Amtagvi has established trial & even better real world data of strong systemic efficacy for 2L post PD1. Delaying this treatment for as you call it “non zero contribution” would be remarkably stupid. Real world 44% ORR/ 52% ORR for true 2L.
English

$REPL
Given keytrudas low ORR in post Nivo setting, we know pd1 contribution is not ZERO but definitely not 30% ORR (as shown by combo).
So both have non zero contribution IMO and while the exact contribution of each component would be “nice to know”, FDA should view this from a patient perspective given how limited options there are in post PD1 setting.
For melanoma patients, this intellectual curiosity matters very little and all they care about is that combo has good activity after failing Pd1.
FDA dropped the ball on this.
English





