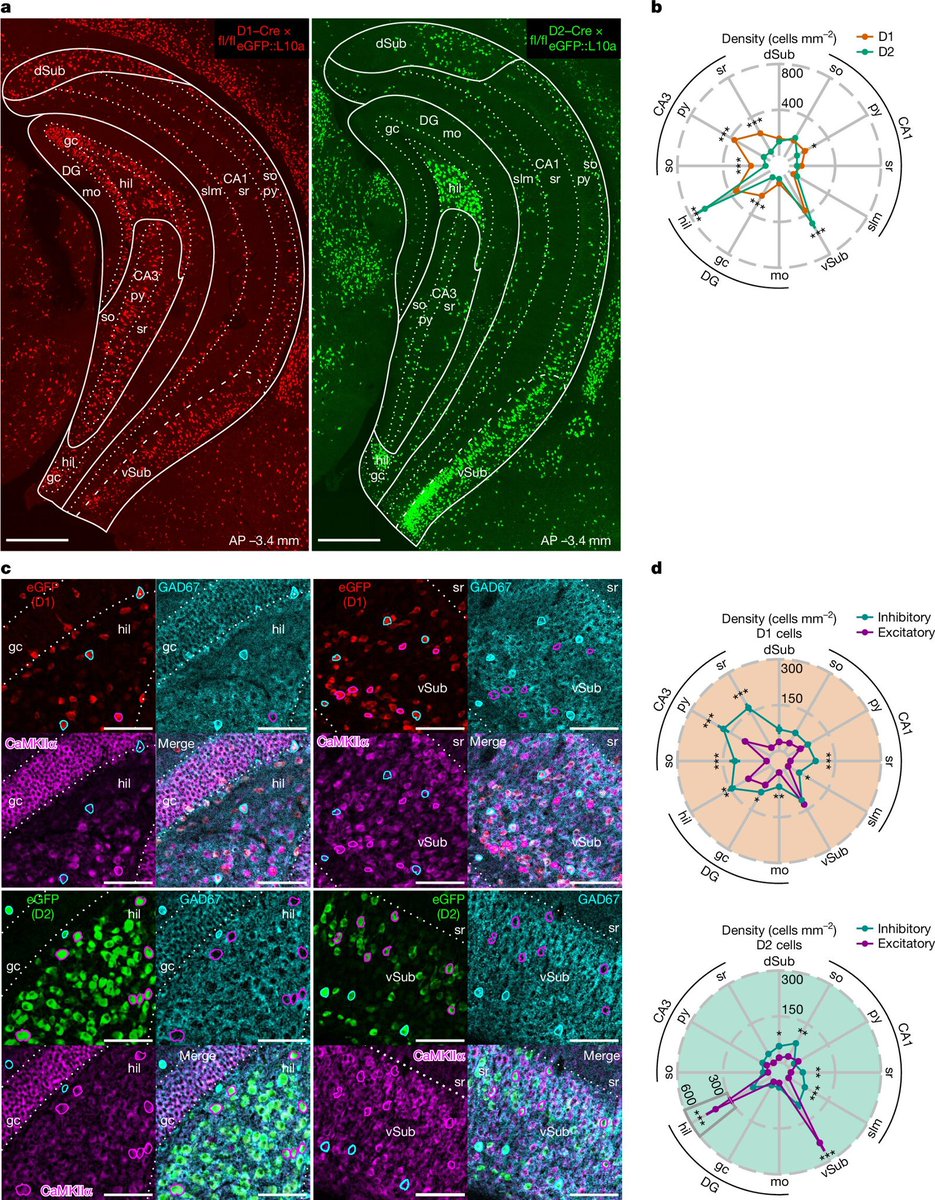
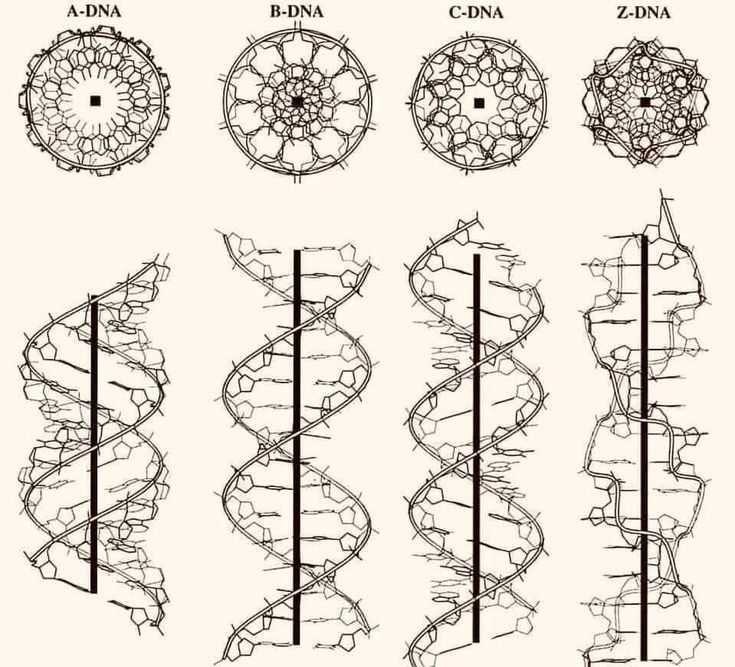
deepTxTF
19K posts


deepTxTF
@3lnahual
against all odds. too old to live, too young too die,



On this day, in 1961, two articles appeared in Nature announcing the isolation of messenger RNA (mRNA) cell.com/current-biolog…

Tal vez no tenemos muy bien dimensionado quiénes son "Los Ardillos". Sus líderes, Carlos Ortega Rosas, alias "La Ardilla", y su hijo Celso Ortega Jiménez, son padre y hermano respectivamente de Bernardo Ortega Jiménez, diputado estatal por el PRD. Además, según los reportes...

Many experiments in biology happen one protein at a time, which means synthesizing DNA one gene at a time. This is fine for tens of genes. For thousands, the cost is unsustainable. Introducing uSort-M: a method to isolate and sequence-verify thousands of genes at low cost








THE GUARDIAN OF THE GENOME: “how P53 stands between us and cancer” The protein p53 —widely known as the guardian of the genome"—emerges as one of biology's most conserved and versatile sentinels, evolved not merely to react to genomic insults but to anticipate and integrate environmental cues that shape cellular fate. Discovered in 1979 as a viral oncoprotein interactor and later recognized for its tumor-suppressive potency, p53 functions as a sequence-specific transcription factor that binds DNA and regulates hundreds of genes. Its activation threshold is tuned by cellular context: under basal conditions, MDM2-mediated ubiquitination keeps p53 levels low; upon stress—DNA breaks from UV, oxidative lesions, oncogene hyperactivation, or redox shifts—ATM/ATR kinases phosphorylate p53, acetyltransferases modify it, and MDM2 inhibition stabilizes it. This allows p53 to enforce genomic integrity by blocking cancer's core hallmarks: unchecked proliferation, death resistance, metabolic reprogramming, and instability. How p53 Prevents Cancer: Core Mechanisms from First Principles p53 operates as a decision node: mild stress favors repair and adaptation; severe or persistent stress triggers elimination. Key outputs include: DNA repair orchestration — p53 induces GADD45 and XPC for nucleotide excision repair (NER), targeting UV-induced cyclobutane pyrimidine dimers and oxidative adducts, preventing fixed mutations during replication. Cell-cycle checkpoints — Transcriptional upregulation of p21 (CDKN1A) enforces G1/S and G2/M arrest, granting time for NER or signaling irreversible exit. Apoptosis commitment— Severe damage activates PUMA, BAX, NOXA, and mitochondrial cytochrome c release, ensuring damaged cells self-destruct rather than propagate errors. Senescence induction — Permanent proliferative arrest maintains tissue architecture while halting mutation accumulation. Metabolic reprogramming — p53 suppresses glycolysis and nucleotide synthesis (curbing Warburg-like proliferation) while promoting autophagy, fatty acid oxidation, and ferroptosis sensitization, aligning energy use with genomic fidelity. These layers create redundancy: no single pathway suffices; failure across multiple axes enables oncogenesis. TP53 mutations occur in ~50% of cancers; indirect inactivation (MDM2 amplification, HPV E6) dominates the rest. Germline TP53 defects (Li-Fraumeni syndrome) confer near-certain multi-tissue cancer risk. p53-knockout mice succumb to spontaneous tumors early; species like elephants with amplified TP53 copies exhibit extraordinarily low cancer rates despite mass and longevity—illustrating dosage-dependent protection. Therapeutic Implications: From Rescue to Preconditioning Pharmacology targets p53 loss reactively (early 2026 status): - MDM2 antagonists (nutlin derivatives, degraders) stabilize wild-type p53 in intact tumors. - Mutant p53 refolders (Y220C binders) or exploiters (synthetic lethality via replication stress, ferroptosis priming). - Prophylactic upregulation in high-risk groups (Li-Fraumeni). - Combinations with immunotherapy, ferroptosis inducers, microenvironment modulators. Yet first-principles demand scrutiny: p53 evolved in light-exposed, seasonally variable, metabolically cyclic environments. It interfaces with circadian clocks, mitochondrial redox, and hormetic stressors. Modern disruption—indoor dim light, constant feeding, absent cold—likely desensitizes p53 thresholds, elevating baseline oncogenic risk. Proactive reinforcement via ancestral cues offers upstream leverage. 1 of 2 T.I.N.A 👑🔆🫀💫 Video credits@nanolive_sa 𝗠𝗲𝗹𝗮𝗻𝗶𝗻 𝗚𝗿𝗮𝗻𝘂𝗹𝗲𝘀 𝗼𝗻 𝘁𝗵𝗲 𝗠𝗼𝘃𝗲 𝗶𝗻 𝗠𝗲𝗹𝗮𝗻𝗼𝗰𝘆𝘁𝗲𝘀 🌟 Watch melanin granules travel within epithelial cells in real time, label-free!