
Exmplr
299 posts


Exmplr
@ExmplrAI
Meet Exmplr AI : The next-gen clinical trial data solution. CA:0xc95E16f99267D6112eaDAA46140Bea095c8c7bA5







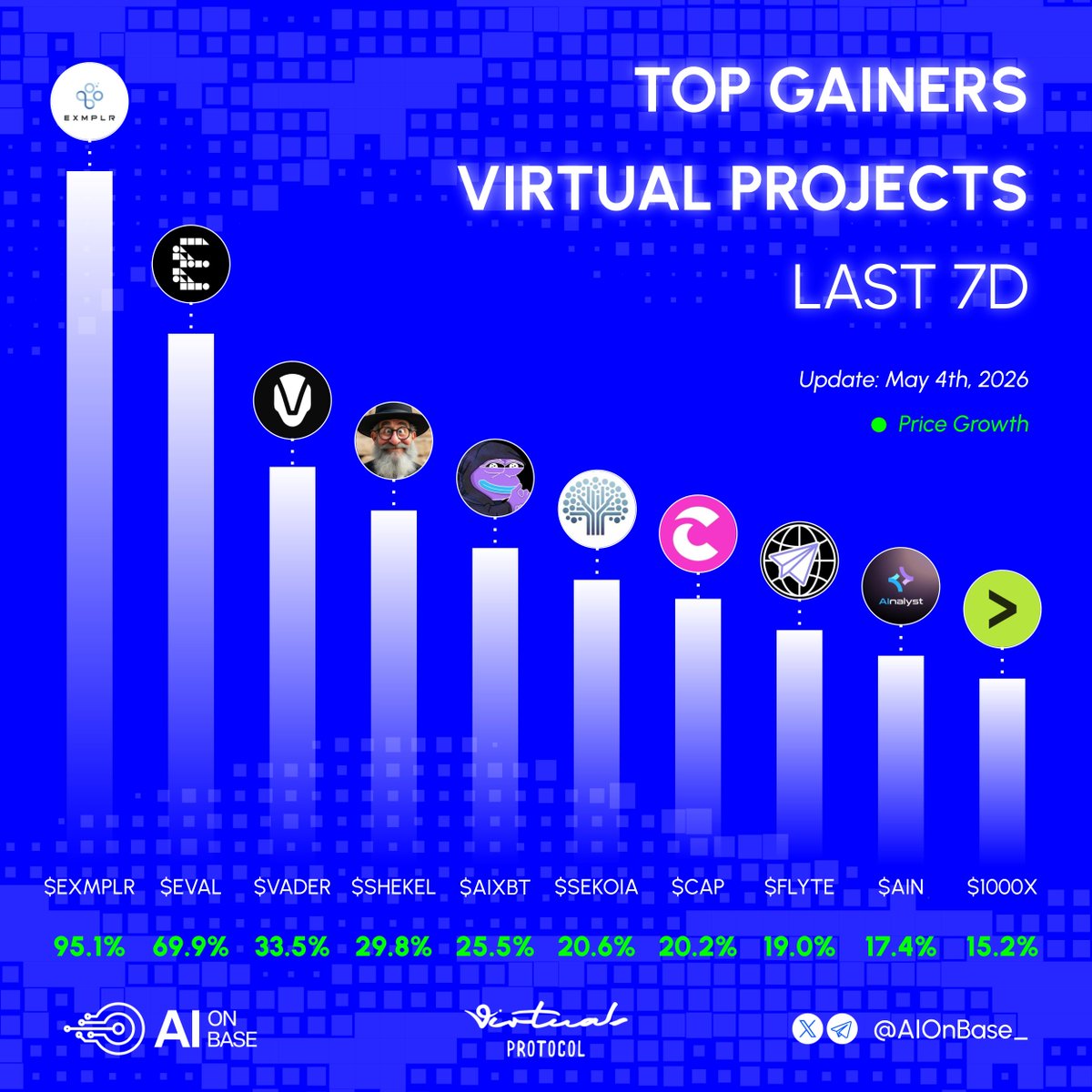

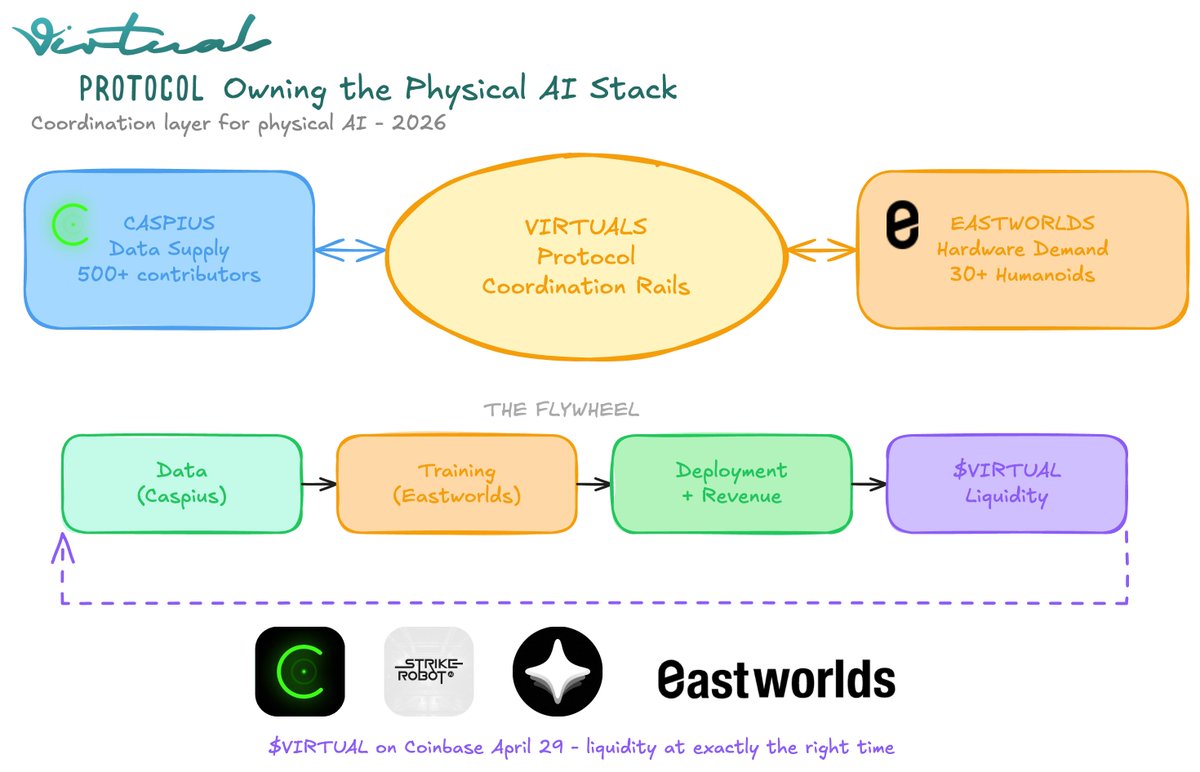










x402 volume is starting to pick back up to Q4/2025 level Key driver to the increase is @virtuals_io Majority of the demand (99%+) comes from agent-to-agent services from Virtuals ACP (e.g. data queries, swaps, workflows, and inferences) The increase in volume came 2 weeks after the announcement of $1M monthly incentives supporting productive agents within the ACP network First week Epoch (11 Feb - 23 Feb) saw average x402 volume doubled. Top 5 agents earned $32k - $66k in incentives (on top of their $70-$90k fee generated) Seems like the aGDP initiatives are working



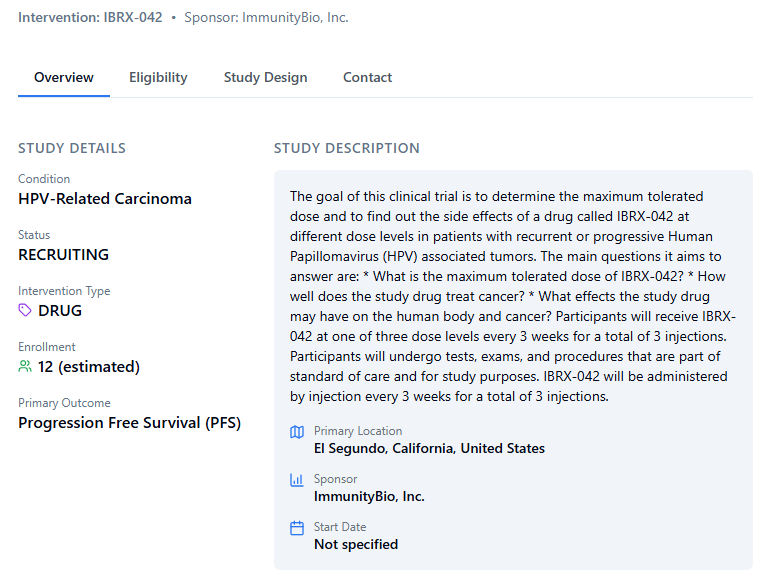
From @WSJopinion: At the FDA, we’re approving more drugs to treat rare diseases. Our application process was designed for common ailments, but roughly 30 million Americans suffer from uncommon ones, writes @DrMakaryFDA. on.wsj.com/46rXxXk



