Gabriel Lazaro-Munoz
901 posts


Gabriel Lazaro-Munoz
@GLMbioethics
Associate Professor @HarvardMed @MGHNeurosurg & @MGHPsychiatry Bioethicist, Neuroscientist, Lawyer. Ethical and social implications of genomics & neuroscience


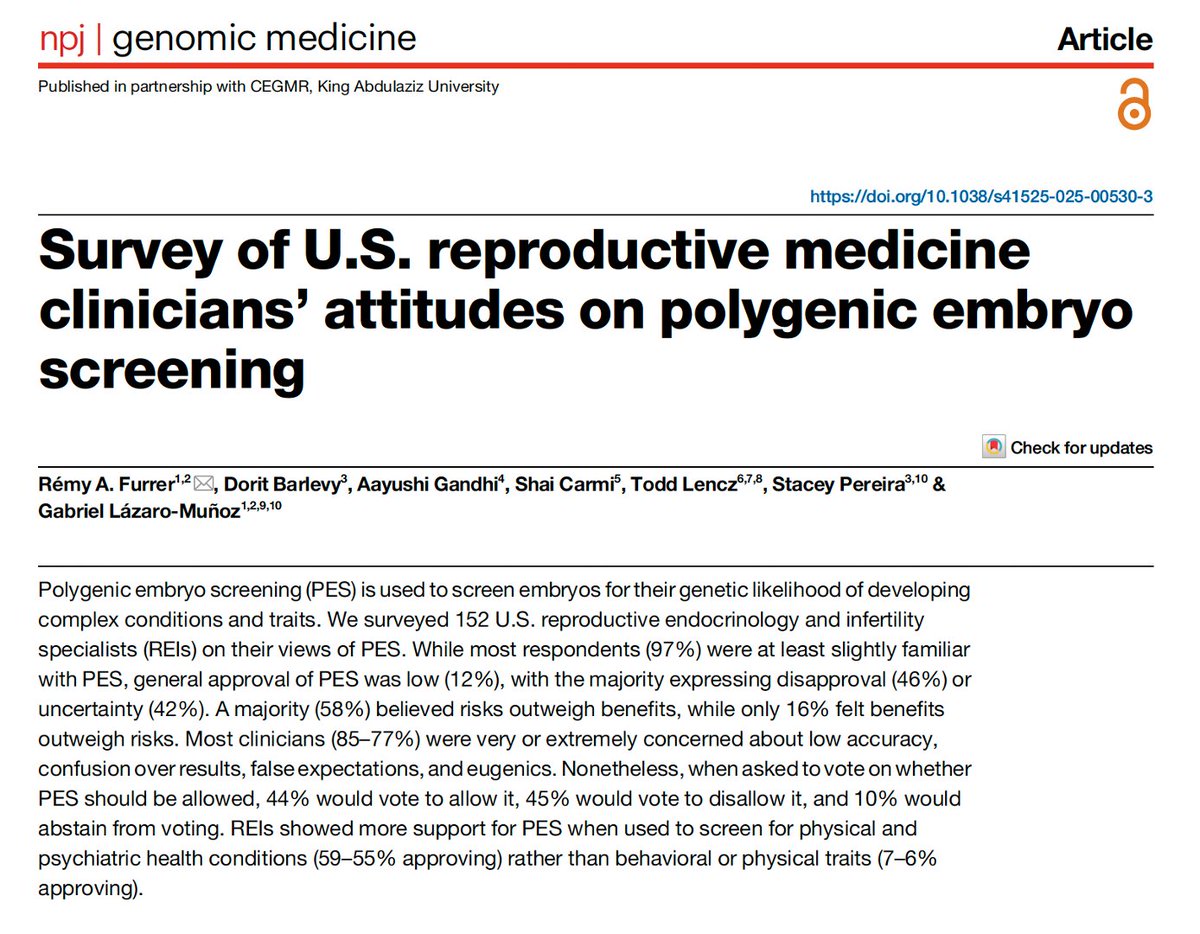
Polygenic embryo screening is being marketed commercially – but how do IVF clinicians view it? • General approval is low (12%) For specific uses: • 59% approved of health-related embryo selection • 6% approved of trait-based selection 🧵 Survey findings in NPJ Genomic Medicine










NJAM Senior Fellows will receive mentorship and training from NJAM Director Dr. Theresa Williamson MD MPH @Twilli7, as well as NJAM Co-Directors Dr. Gabriel Lázaro Muñoz PhD JD @GLMbioethics and Dr. Francis Shen JD PhD dana.org/article/neurot…







What happens if you have a device implanted in your brain and the company goes out of business? What happens if the device was in trial and is not FDA approved? What happens when a company cannot support the product? Out today in @JAMANetworkOpen new consensus on device abandonment inspired by @royalsociety meeting in Sept. 2023. Key Points: - This paper provides a definition of implanted neurological device abandonment. - It also provides a systematic review and consensus statement. - 734 articles published only 7 were relevant to neurological device abandonment. - A multistakeholder group developed a consensus definition for neurological device abandonment. - The definition included 'devices used in deep brain stimulation, vagal nerve stimulation, and spinal cord stimulation, including failures related to patient consent, support before the end of the device's lifespan, and safety concerns.' My take: We need to be proactive as the neurological device community inevitably grows, abandonment will become more and more common. There is thus, a huge looming problem emerging in the field of brain and spinal cord implants and there will be device abandonment. Establishing a formal definition of neurological device abandonment will be critical for development of guidelines, policies, and laws to reduce or prevent such abandonment. The definition for neurological device abandonment should 'entail failure to provide fundamental aspects of patient consent; fulfill reasonable responsibility for medical, technical, or financial support prior to the end of the device’s labeled lifetime; and address any or all immediate needs that may result in safety concerns or device ineffectiveness and that the definition of abandonment associated with the failure of a research trial should be contingent on specific circumstances.' We need to have the dialogue on abandonment today and not 'kick the can down the road,' which could trigger critical safety and access concerns. jamanetwork.com/journals/jaman… #deepbrainstimulation #neuromodulation #DBS #Parkinsons

The Deep Brain Stimulation (DBS) Think Tank XI proceedings is published! “Pushing the Forefront of Neuromodulation,” the updates, paper and meeting were highlighted by @ndosenbach who presented research recently published in @Nature w/Evan Gordon to identify and characterize the somato-cognitive action network (SCAN), which has redefined the motor homunculus and has led to new hypotheses about the integrative networks underpinning therapeutic DBS. frontiersin.org/articles/10.33… #Parkinsons @DBSThinkTank @andreashorn_ @leaddbs @foxmdphd @HelenMaybergMD @JWongggMD #deepbrainstimulation #neuromodulation