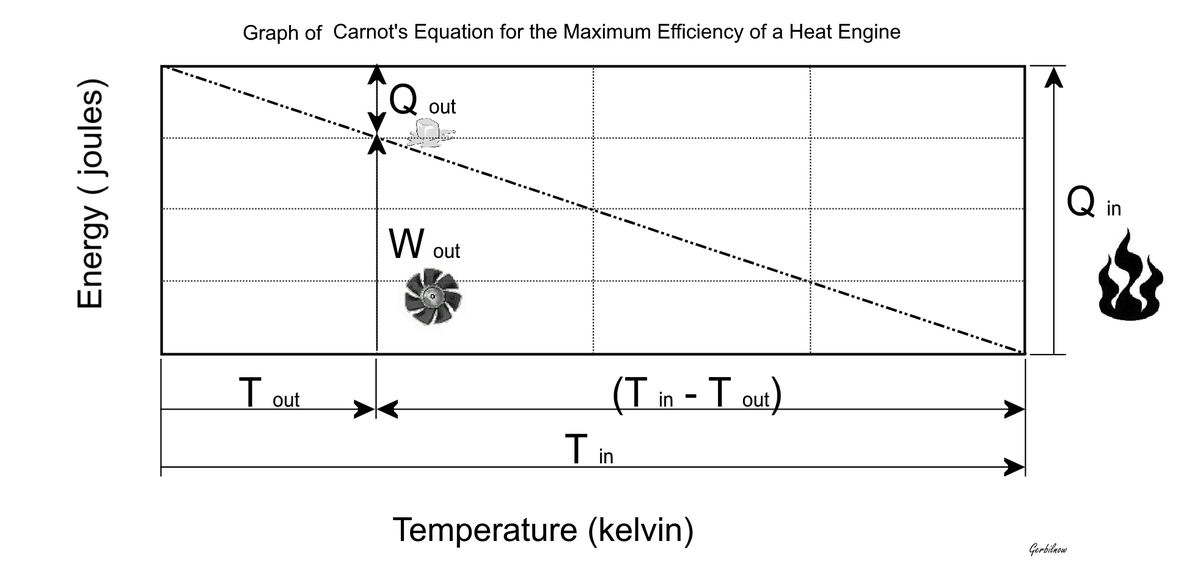
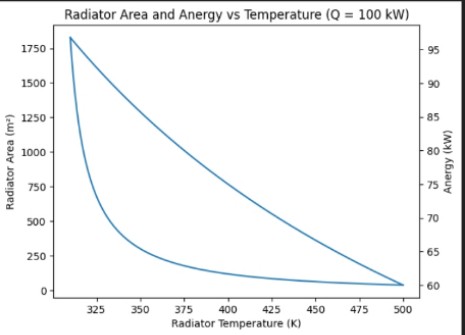
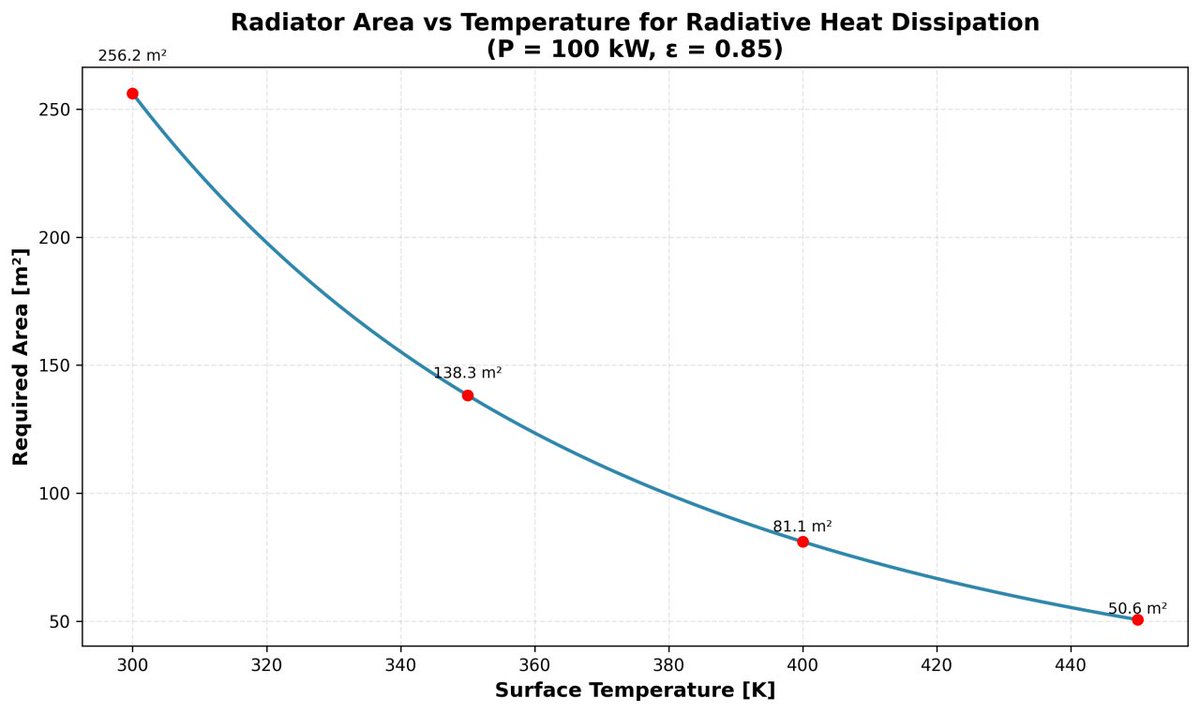
@LaloMedecigoMR WOW nunca nos contestaron ni por aquí ni por face ni por las páginas oficiales @LaloMedecigoMR ni modo
Español
Rocktor Cobos
5.8K posts


@JACobosMurcia
Doctor en Ciencias; Químico por Vocación, Rockero por Pasión, Tornero por Admiración, Fotografo por Decisión y Cocinero por Sabor: Cosamaloapeño errante (Q≤T∆S)