Sabitlenmiş Tweet

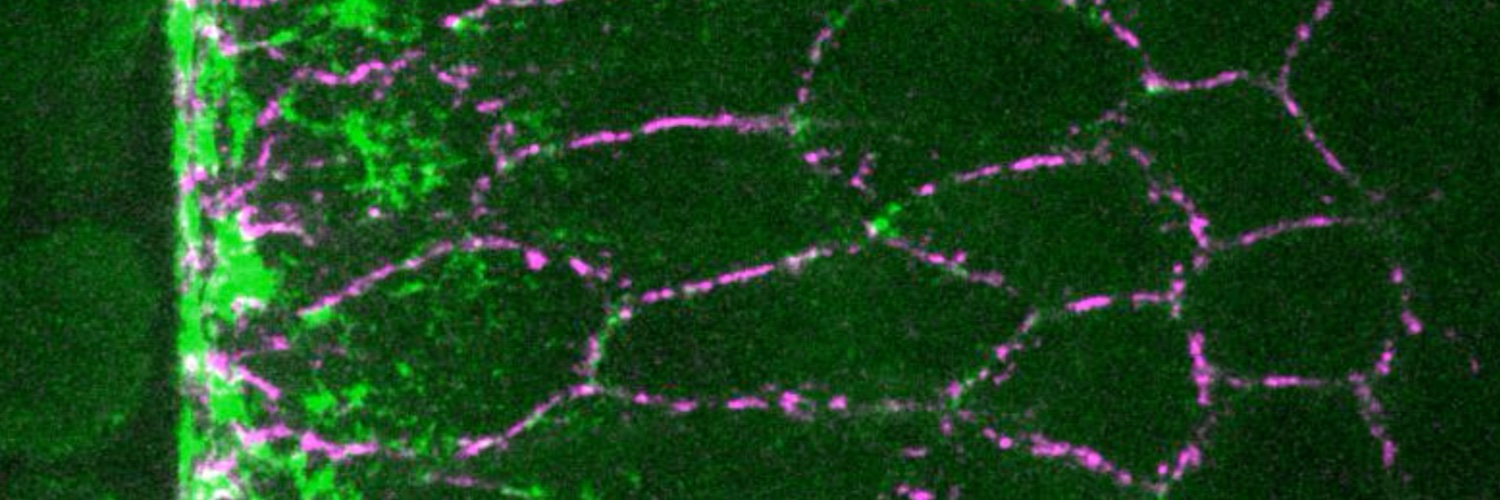
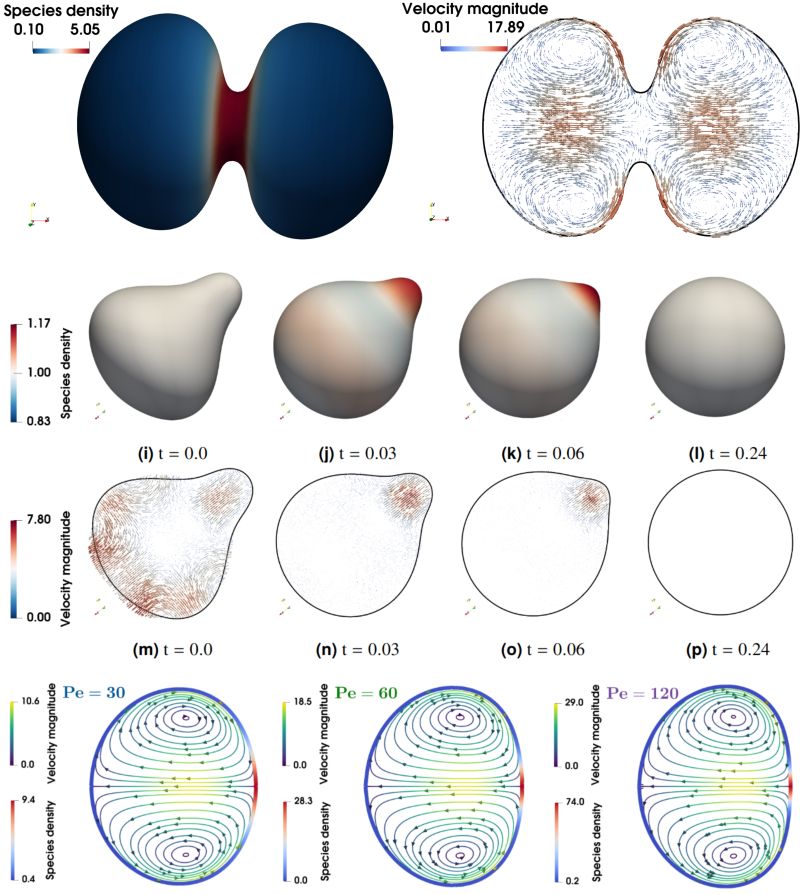
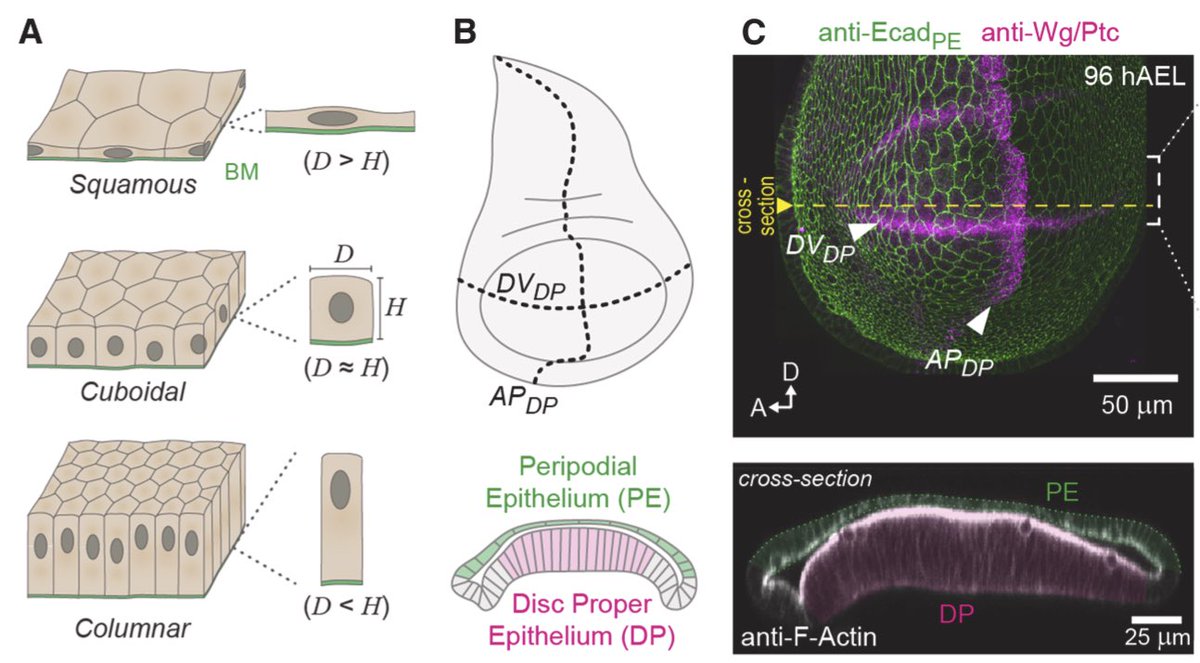
Here is video of 1st lecture at College de France on Biological computation 😊. I defend the necessity to access Algorithmic & Computational levels of analysis in cell & dev. biol. proposed by David Marr in neuroscience. Logic & Function matter 🤩.
Video: tinyurl.com/55yejk5f

English