
cpamtm
347 posts


cpamtm
@cpamtm
Management Consulting and Advisory







From today, the @MHRAgovuk-NICE Aligned Pathway is open, delivering parallel decisions on licencing and value to get medicines to NHS patients 3-6 months sooner. No separate application or fee needed for eligible topics. Learn more: nice.org.uk/news/blogs/mhr…


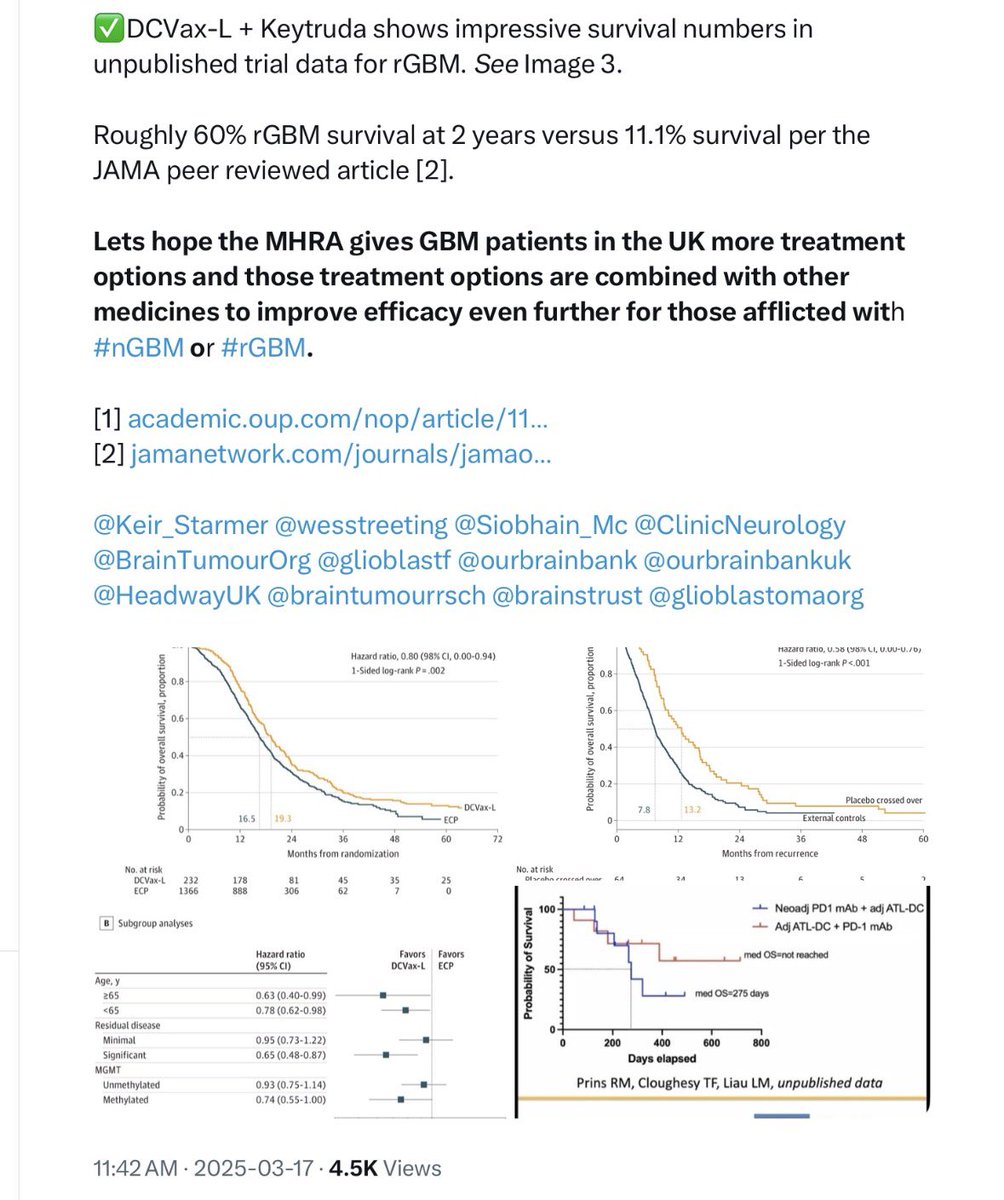
NWBO DCVAX-L @MHRAgovuk @NHSuk @UKParliamentNow In 832 days you have not approved a treatment platform that would greatly benefit over 6000 UK Citizens annually. This slow walking is impacting quality of life for those in need of it and holding back other treatments in the pipeline. A conditional approval should be given as you have allowed it to be used for years through compassionate use specials programs. 832 days and no approval shows that the process is broken especially given it is allow to be used via the specials programs for over 900 patients of which 300+ were xGBM treatments. By slow walking this it appears there is corruption somewhere in the process as well. Leadership should be investigating why there teams are moving so slow. How many more UK citizens have to die while you all noodle on the approval.






Phil Woolas, Littleborough and Saddleworth MP 1997- 2010 passed away last night. A lovely man&very able colleague. Last time I saw him was June 2024 at the Glioblastoma Drug trial fundraiser in memory of my sister. Last night the same disease took his life. We will find a cure!




Based on the history of the #MHRA approval process it should've been approved already (or not approved), but if it was to be denied my understanding is we would've known a while ago, so I get the "Imminent" belief. But I agree it's annoying listening to the predictions come and go.














The National Cancer Plan will transform care for rare cancer patients. We're investing in research, appointing a new national lead and helping patients find clinical trials faster. @KarinSmyth visited St James's University Hospital in Leeds to see the @NIHRresearch facility.







