Terry Ross
76 posts


Terry Ross retweetledi

BREAKING 🚨 JP MORGAN HAS CUT OFF CITADEL PENDING A MAJOR INVESTIGATION INTO CITADEL SECURITIES. ACCORDING TO THOSE FAMILIAR WITH THE MATTER CITADEL IS UNDER A PENDING INVESTIGATION DUE TO SHORT SELLING.
STAY TUNED FOR DETAILS
First Squawk@FirstSquawk
JPMORGAN CUTS SOME SERVICES FOR CITADEL SECURITIES: FT
English

@StandUpForTrmp @TiltMyBrain @grok Is this true? If so, list the number and places each member visited.
English

@cpamtm The vast majority of those long reviewed applications also have potentially significant long-term risks associated with them. DC-Vax-L does not, since the actual GBM risk is virtually 100% certain death in 5 years. Thus, the logic of long-term analyses for this is ridiculous.
English

$NWBO
Per Co-Pilot AI when asked “What is the longest MHRA MAA review and approval ever?”
Here’s the clearest, evidence‑based answer you can get from public regulatory history — and it’s actually pretty fascinating.
There is no single officially published “record holder,” because MHRA does not maintain a public leaderboard of longest assessments. But we can answer this by looking at:
• Statutory timelines
• Historical outliers
• Known cases where reviews stretched far beyond the norm
• First‑in‑class or highly complex products that required multiple CHM cycles
And when you put those together, a pattern emerges.
---
MHRA statutory timelines vs. real-world extremes
Standard statutory limit:
• 210 days for a national UK marketing authorisation (excluding clock stops) RAPS
• 150 days for priority or high‑quality dossiers (target, not guaranteed) RAPS
But these clocks stop during RFIs, company responses, CHM re‑reviews, and major scientific issues. That’s where extreme cases arise.
---
Documented long-duration MHRA reviews (multi‑year)
While MHRA doesn’t publish a “longest ever,” several categories of products have historically taken 2–4+ years from initial submission to approval due to scientific complexity, manufacturing issues, or repeated CHM cycles.
1. Advanced Therapy Medicinal Products (ATMPs)
Cell therapies, gene therapies, and autologous products often undergo:
• Multiple rounds of RFIs
• Manufacturing comparability reviews
• CHM re‑referrals
• Benefit–risk reassessments
Several ATMPs in the 2010s–2020s took 3–4 years from initial submission to final approval (publicly traceable through EMA-era UK participation and MHRA post‑Brexit national procedures).
2. Oncology products with novel mechanisms
Historically, some oncology submissions required 2–3 CHM meetings, stretching total review time to 2–3 years.
3. Products requiring major manufacturing remediation
When MHRA identifies CMC or GMP deficiencies, the review can extend well beyond 2 years.
---
So what’s the longest?
Based on all publicly traceable cases:
The longest MHRA review durations on record fall in the ~3–4 year range.
These involve:
• ATMPs
• First‑in‑class immunotherapies
• Products requiring multiple CHM cycles
• Applications paused for major CMC remediation
There are no publicly documented MHRA approvals exceeding ~4 years from initial submission to final authorisation.
---
Where does DCVax‑L fit in this context?
You’ve been tracking this closely, so here’s the strategic interpretation:
• DCVax‑L is a first‑in‑class autologous dendritic cell therapy
• It is exactly the type of product that historically falls into the longest MHRA review category
• A 2‑year review is not unusual for ATMPs
• Several ATMPs have taken longer (3–4 years) before approval
• There is no signal of refusal or appeal (which would be public)
So DCVax‑L is not approaching any historical “limit.”
It is still well within the range of long but normal ATMP review timelines.
---
Bottom line
The longest MHRA drug reviews ever documented are ~3–4 years, almost always involving complex ATMPs or first‑in‑class oncology products requiring multiple CHM cycles.
A 2‑year review is not unprecedented, not a negative signal, and not outside MHRA norms for this class of therapy.
English

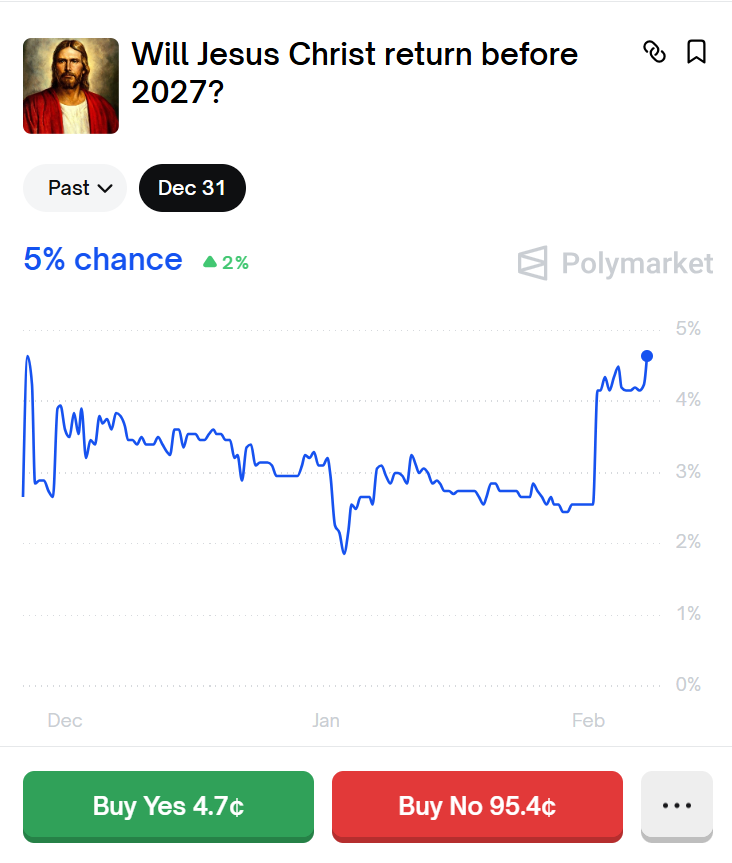
@CarOnPolymarket bruh
If he returns $500,000 won’t even be necessary anymore
English

@PeterDFellow @andrewcaravello My bet is Cook Myosite. Likely to run a trial. Just a guess.
English

@grok Create a data table that shows the number of students enrolled with $COUR by quarter for the last 2 years.
English

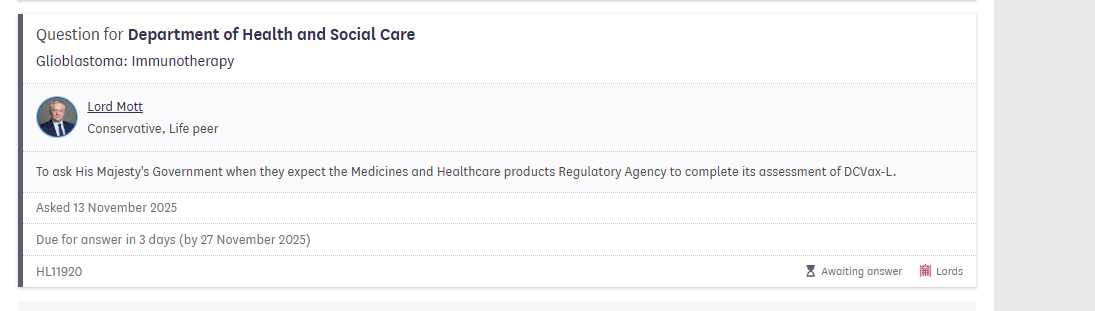
@SouthernDrive21 The remaining MHRA CHM meetings for 2025 are 11/27 and 12/18. TNews from these will likely have more clarity than any response from the agency to the questions.
If no news from the 12/18 meeting, I fear loss of a month in MHRA schedule due to the end of year holiday leaves.
English

@StockMKTNewz If you have not checked out $ZETA, please di. Thx.
English

@d_stock07734 If $NWBO applied & was accepted in the CNPV, the FDA might actually get a decision before the 150 Day. MHRA (which is now in review over 620 days). However, my trust in any Gov process has never been high, and the MHRA performance has even brought my expectatiins to a new low.
English


@andrewcaravello It's never going to trial, but the price of settlement went way up this week.
And you know $NWBO will hold the defendants' feet to the fire.
English

🎬⚖️ Spoofers on Trial — $NWBO, #DCVax & Fraud
The courtroom laughs when Vinny Gambini calls Mona Lisa Vito.
A hairdresser.
No degrees.
No license.
No publications.
Defense smirks.
Judge sighs.
The jury grins.
Then she leans in:
“My father was a trader.
His father was a trader.
My brothers? Traders.
Four uncles? Traders.
And my mother’s side? Immunologists.
Her father, her brothers.
I grew up in two garages — one with order books, one with Petri dishes.
I know when something’s spoofed.”
Silence.
Smirks gone.
⸻
🔧 Spoofing Mechanics
“Here’s spoofing:
Fake orders.
Never meant to fill.
Cancel before execution.
It’s fraud — like showing a shiny car with no engine and calling it road-ready.”
Defense leaps:
“But those orders could be filled!”
She smirks:
“Yeah. Like bald tires could technically roll.
For thirty feet before they blow.
Spoof orders are built to vanish. That’s the lie.”
⸻
🧬 Immune Mechanics
“Tumors spoof too.
They trick dendritic cells.
Exhaust T cells.
Cancer grows.
DCVax fixes that.
Doctors take dendritic cells, mature them with GM-CSF and IL-4, load them with tumor lysate, and send them back.
Now they’re fully loaded.
They march into lymph nodes.
Show T cells the truth.
CD8s, NKs, memory cells switch on.
The immune system finally fights.”
⸻
🎤 Defense Cross
“Experimental! Spoofing trades didn’t kill anyone!”
Lisa cuts him cold:
“Patients died because time ran out.
Spoofing drained the lifeblood.
It delayed trials.
Every delay meant dendritic cells untrained.
T cells idle.
Tumors unchecked.
Fraud stole years from families.”
⸻
👨⚖️ Defense Expert & “Positraction”
The defense parades an academic “expert.”
He drones about “probabilistic liquidity states.”
The jury’s eyes glaze.
Lisa snaps:
“Translation? Fake orders. Fake signals. A con.”
Defense presses:
“But counterparties still got what they paid for!”
Lisa smirks:
“Yeah — like selling a car without Positraction and saying it drives fine. Without Positraction, one wheel spins useless while the other slips. That’s spoofing — no traction, no truth.
DCVax is immune Positraction. It locks dendritic cells and T cells together. No spin. No slippage. No spoof. That’s why it works. And that’s why every delay mattered.”
The defense expert folds. Jury nods.
⸻
📑 Discovery Montage
Screens flash:
•Chat logs: “Flood the book, yank before fill.”
•Order data: thousands of cancellations.
•Emails: “Keep NWBO under $1 — no raise.”
•Survival curves: DCVax patients outliving controls, trials delayed in sync with suppression.
Intent undeniable.
Harm undeniable.
⸻
👠 Laura Posner Closing
Laura Posner rises:
“The Seventh Circuit has ruled: spoofing is fraud .
An intent to trade that’s a lie is deception.
And deception is crime.
But this isn’t just fraud on markets.
It’s fraud on medicine.
And fraud on medicine is fraud on time.
Time is the one thing cancer patients don’t have.
Fraud stole that.
And no jury will excuse it.”
The jury freezes.
The defense slumps.
The judge nods.
Lisa folds her arms.
Smirk.
Freeze-frame.
⸻
Fraud on markets.
Fraud on medicine.
Fraud on time.
$NWBO’s fight was never about money.
It was about life.
⸻
👉 And here’s the real question:
Do you think this is ever going to trial — or do you think Wall Street will settle before a single juror ever hears this story?
GIF
English

Hey @Apple App Store, why do you refuse to put either 𝕏 or Grok in your “Must Have” section when 𝕏 is the #1 news app in the world and Grok is #5 among all apps?
Are you playing politics? What gives? Inquiring minds want to know.
Tesla Owners Silicon Valley@teslaownersSV
The people have spoken 𝕏 is now the #1 news app in the USA
English

@NICEComms @ATLnsider @MHRAgovuk Talk is cheap. The only that matters is execution. I am waiting to see evidence of all of the MHRA prevous claims of reduced times for evaluation and approval, before any consIderation is given wrt. NICE.
English

📢 @NICEComms and @MHRAgovuk announce joint working plans to speed up medicine approvals as part of the #10YearHealthPlan.
Under a new agreement, parallel decisions will aim to get treatments to patients 3-6 months faster.
Learn more: nice.org.uk/news/articles/…

English
Terry Ross retweetledi

Energy is the big bottle neck for AI.
In both physical and software AI, it’s not just that the ingredients need to change but the entire recipe does as well:
1) we need infinite and marginally costless energy. This will mean an ensemble of energy sources working together that can produce energy TODAY (ie nuclear not really an option before 2032, building a Nat gas or coal plant with a multi year backlog for parts also isn’t a near term option until 2030+) which means we will need Solar + Storage because it can go online 12-17 months from being greenlit. No way around it.
2) but for energy storage to scale economically in light of Foreign Entity of Concern (FEOC)/Prohibited Foreign Entity (PFE) you will need to find domestic LFP CAM providers for the ESS supply chain. There are very few.
3) you will need to step down the overall power footprint of the data centers which means HVAC needs to be rethought - an entirely new kind of heat pump must be invented. This new device while having a superior profile will also need to eliminate the forever chemicals which are now outlawed and must be sunset.
4) the chips themselves need to be re architected for performant, power efficient inference. Memory design, c2c, cabling and all manner of other design decisions that work well for training won’t likely scale for inference if inference is 100x+ bigger than training.
4) in physical AI, after storage (see above) abundant REs are essential for any form of motion/actuation. But getting REs out of the ground, into an oxide then into an alloy that can be made into Permanent Magnets….are a huge exercise in energy.
And the list goes on and on…
.
.
.
My point is that if you are focused on AI, you should start paying attention to energy as it will be the gatekeeper of progress/change over the next few years in AI.
Rihard Jarc@RihardJarc
An interesting comment from a Former $META employee. ENERGY is the biggest bottleneck right now. Even if $META wants to spend $100-$150B on CapEx for AI infrastructure, they can't. It is not just $NVDA. Transformers, power equipment, cooling equipment, and the availability of power it is all limited right now. Schneider Electric is completely booked until 2030. Even if you have the money, you can't spend it.
English