ATHANASIOU DIMITRIOS retweetledi

We waited far too long for the results of the For-DMD study, but here they are: jamanetwork.com/journals/jama/…
English
ATHANASIOU DIMITRIOS
50 posts


@dathax
Duchenne Patient Advocate PDCO member in European Medicines Agency, Board Member of World Duchenne Organization, Board Member of EPF










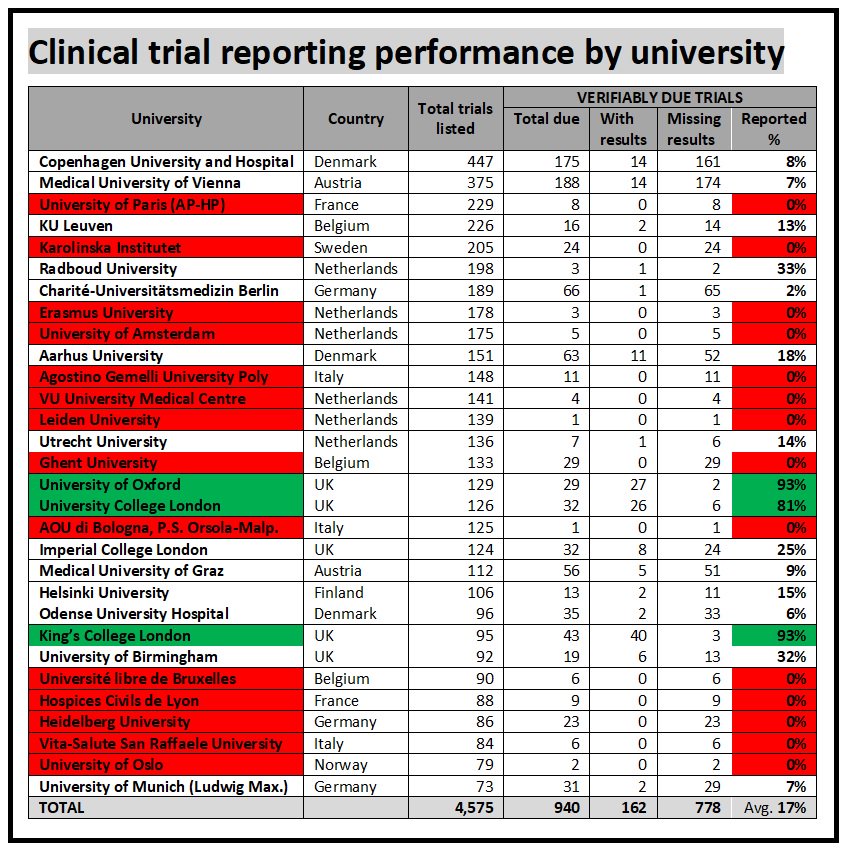






@ReveraGen release details of vision-dmd phase 2a study vision-dmd.info/vision1/wp-con… Eric Hoffman will present @ParentProjectMD Pls RT @worldduchenne @trinds_nmd @TREAT_NMD




