Pawan Rao MD, FASN🩺 retweetledi

AACE 2026 – Type 2 Diabetes Algorithm Update
🔹 Pearl 1 — Diabetes care is no longer glucose-centric
AACE 2026 shifts the paradigm from HbA1c control to comprehensive cardiometabolic risk reduction.
🔹 Pearl 2 — Obesity is now a primary therapeutic target
Weight management is not adjunct—it is central to both prevention and treatment of T2D.
🔹 Pearl 3 — Treat adiposity early, not late
Early use of anti-obesity pharmacotherapy (e.g., GLP-1 RA / dual incretins) is encouraged to alter disease trajectory.
🔹 Pearl 4 — Complications drive therapy selection
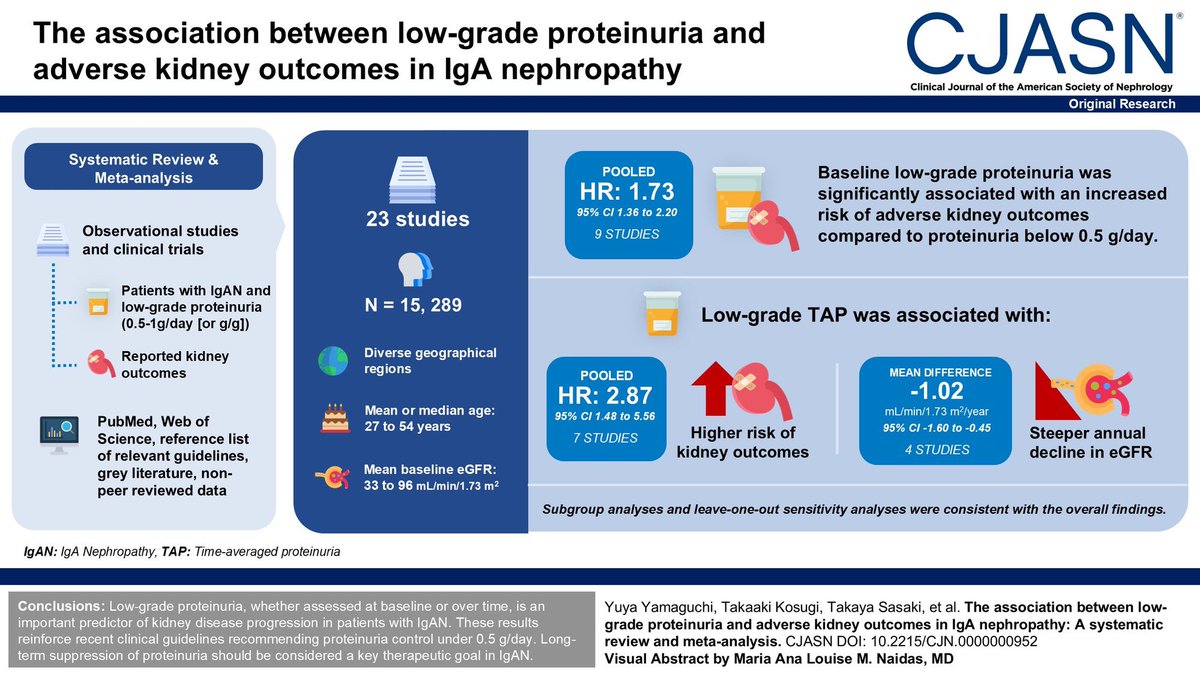
Choice of drugs should be guided by ASCVD, CKD, HF, and obesity—not just HbA1c level.
🔹 Pearl 5 — GLP-1 RA and SGLT2 inhibitors remain foundational
These agents are prioritized for both glycemic control and organ protection (heart, kidney, weight).
🔹 Pearl 6 — “Right diagnosis” precedes “right treatment”
AACE introduces emphasis on evaluating diabetes type—avoid mislabeling all patients as T2D.
🔹 Pearl 7 — Prediabetes is a treatable disease state
Aggressive lifestyle + weight-focused interventions can delay or prevent progression.
🔹 Pearl 8 — Dyslipidemia management is integral, not optional
LDL-C and non-HDL targets are critical components of diabetes care.
🔹 Pearl 9 — Hypertension control is part of diabetes therapy
Blood pressure optimization is a core pillar of reducing macrovascular risk.
🔹 Pearl 10 — Personalization is the new standard
Therapy must be individualized based on phenotype: obesity, sarcopenia, CV risk, renal status, and age.
🔹 Pearl 11 — Move from “treat to target” to “treat to outcome”
Focus shifts toward reducing events (MACE, CKD progression, mortality), not just numbers.
🔹 Pearl 12 — Diabetes is a multisystem disease
Management must integrate metabolic, cardiovascular, renal, and functional domains.
🔶 CME INDIA One-Line Takeaway
“AACE 2026 redefines diabetes care: treat weight early, reduce risk aggressively, and individualize therapy beyond glucose.”
endocrinepractice.org/article/S1530-…

English