Brian Grimm, M.D.@DrGrimmMD
Your Office Lights Might Be Making Your Diabetes Worse. Should we start filing for “Workman’s Compensation?”
I’m not trying to be dramatic, but a new randomized crossover study in Cell Metabolism showed something that should make all of us pause. People with type 2 diabetes were placed in two nearly identical office environments for 4.5 days. Same meals. Same sleep timing. Same activity. The only thing that changed was the lighting. One condition used typical artificial office light around 300 lux. The other used natural daylight through windows. Not outside sun exposure. Just daylight coming through glass.
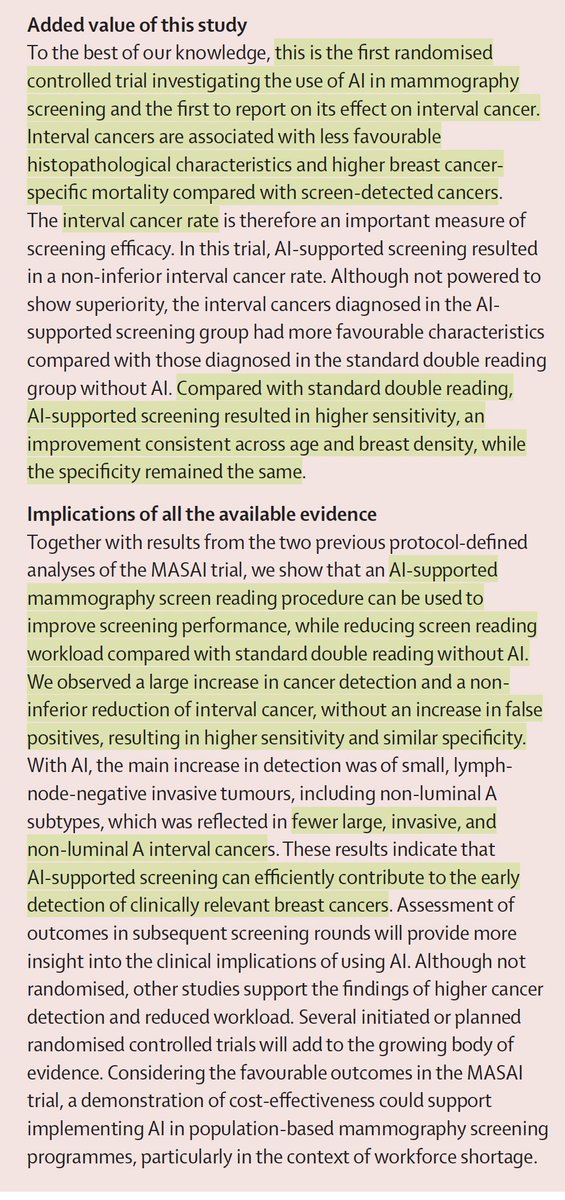
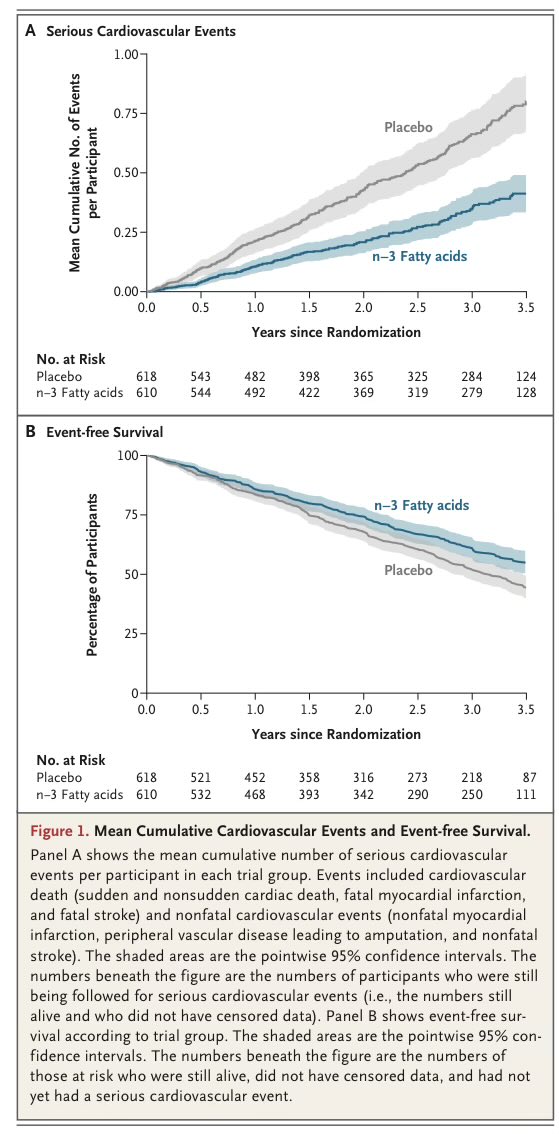
The results were not subtle. Under natural daylight, participants spent more time in the normal glucose range. Their whole-body metabolism shifted toward greater fat oxidation. Evening melatonin output was higher. Skeletal muscle clock genes shifted phase. Ceramide-related lipid signatures, which are strongly linked to insulin resistance, moved in a favorable direction.
Nothing about calories changed. Only the light changed. And in such a short period of time.
That matters because glucose stability is not just about carbohydrate intake. Time-in-range is tied to long-term outcomes and mortality risk in diabetes. In this study, natural daylight increased time spent in the normal glucose range without altering medication or diet. It also reduced the amplitude of 24-hour glucose swings. That suggests improved circadian coherence, not just a transient metabolic blip.
Now here is where it gets deeper. Natural daylight increased evening melatonin levels. We have been mistaken that melatonin is JUST a sleep hormone.
It regulates mitochondrial function, redox balance, autophagy, and apoptotic signaling. It stabilizes healthy cells and helps damaged ones exit cleanly. Ceramides, on the other hand, promote insulin resistance and inflammatory signaling. The fact that daylight exposure was associated with both stronger melatonin output and lower ceramide signatures is not random. That is metabolic timing interacting with cellular quality control.
For those of you who have read John Ott, this should feel familiar. Decades ago, Ott showed that animals and plants raised under full-spectrum light behaved differently than those raised under narrow artificial light. Their growth patterns changed. Tumor behavior changed. Reproductive cycles changed. He was not talking about brightness. He was talking about spectral completeness. Filter the spectrum, change the biology. Something to remember.
This study did not even use outdoor sun. It used daylight filtered through glass, which blocks UVB/A and attenuates portions of infrared. And it still shifted clock gene expression, lipid signaling, and substrate selection. Imagine what would happen if participants had stepped outside mid-morning and mid-afternoon. Imagine layering in UVA nitric oxide release and near-infrared mitochondrial stimulation. The effect we are seeing here may be a conservative estimate.
Here is the practical question. How many people with metabolic disease spend eight to ten hours a day under static LED panels and then try to fix everything with diet and medication? We redesign offices for posture and productivity but rarely question the spectral environment. Yet the retina sets the central clock. The clock sets metabolic timing. And metabolic timing influences mitochondrial efficiency and lipid handling.
You can count macros. You can tweak medication. But if your light environment is misaligned, you are swimming upstream.
We have to stop thinking of Light a decoration in the room. It is YOUR metabolic instruction manual.
Doc, why should you care? Because the built environment may be nudging glucose variability and mitochondrial stress every single workday. And it is a lever most clinicians are not even thinking about yet.
Field Medicine says: fix the signal first.