
Victor Robert
55.1K posts


Victor Robert
@viktorobert
Humanist, Technologist, Free Thinker, 3X founder. Fusion, AI and robotics will usher in humanities golden age. We will rise to the ocean of stars



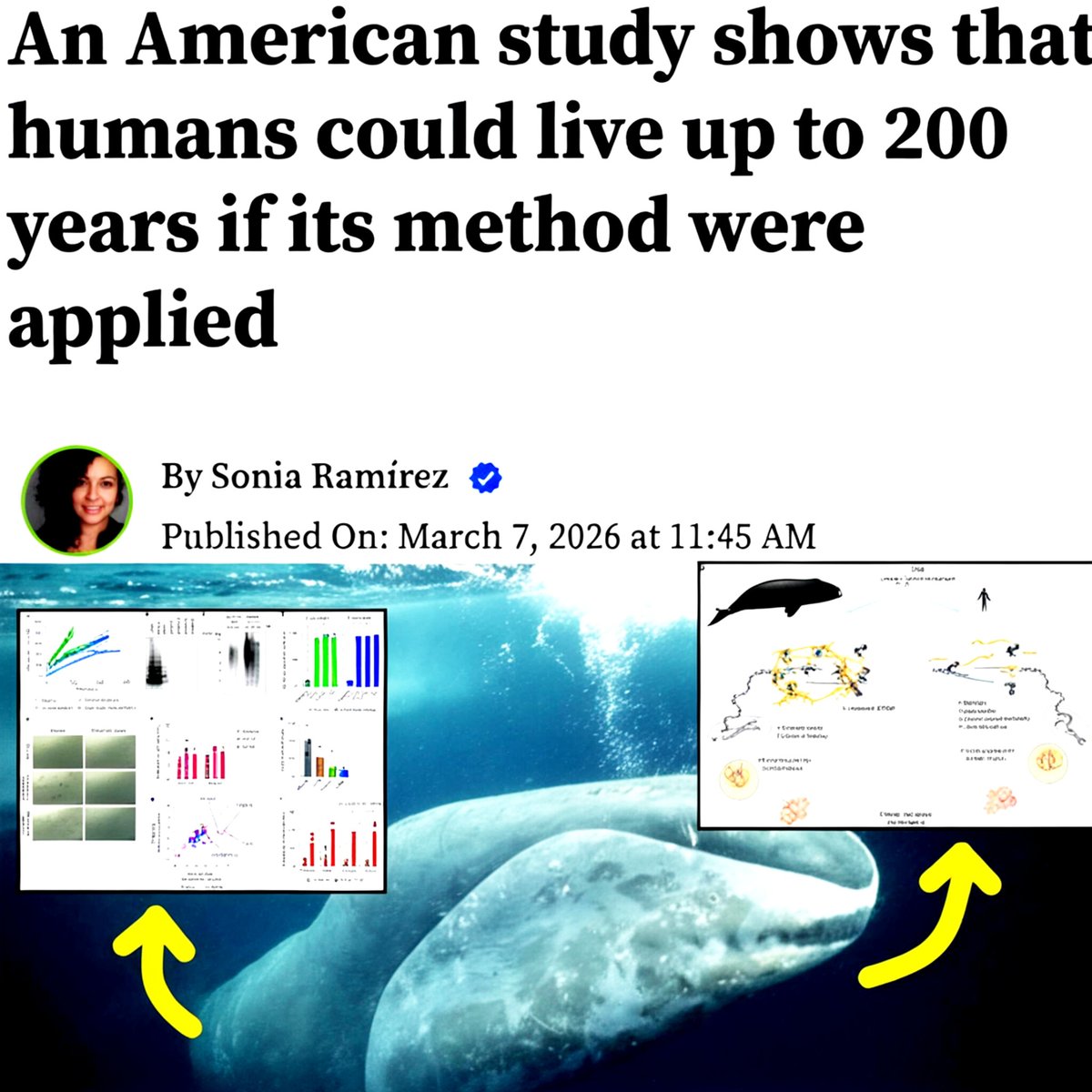
Longevity begins where DNA repair outruns DNA damage. Scientists at the University of Rochester discovered that bowhead whales, which can live over 200 years, produce extremely high levels of a DNA-repair protein called CIRBP (Cold-Inducible RNA-Binding Protein). This protein improves the repair of dangerous DNA double-strand breaks, one of the main sources of mutations, cancer, and aging. When researchers introduced the whale version of CIRBP into human cells, DNA repair became more efficient. In fruit flies, increasing CIRBP levels extended lifespan and improved resistance to radiation damage. The findings suggest that strengthening DNA repair mechanisms could help slow aging and potentially extend healthy human lifespan in the future.





Intel Foundry unveils the world’s thinnest GaN chiplet (19 μm). By integrating power and digital control on a single chiplet, it delivers higher efficiency, faster switching, and smaller designs. Learn more: ms.spr.ly/6018Q2h90 #IntelFoundry #Semiconductors







🚨 BREAKING: Scientists reversed the age of human cells by 30 years. 53-year-old cells… turned into 23-year-old ones. That shouldn’t be possible. Unless aging isn’t time passing… but systems drifting out of balance. And now we’ve figured out how to push it back. If time isn’t what we think… what else are we getting wrong? Follow this gets deeper.

#ICYMI: Honored to be on @Nasdaq’s Tower celebrating our fully subscribed $80M Series D financing supporting our Phase 1 clinical trial & Partial Epigenetic Reprogramming platform. Thank you to our co-founders, Dr. @DavidASinclair, AO & Tristan Edwards. bit.ly/3PUqVA1









