David Din, CFA
2.1K posts


David Din, CFA
@DavidDin
Investor in PLTR, IBRX, ONDS and EOSE. Former banker and buy-side analyst at UBS . I care about Alzheimer and alternative medicine.










30年前くらいに村上春樹のエッセイで、アメリカではコーラにピーナッツを入れて飲むのがポピュラーだと書いてあった。「ふぅん」と思ってから長い時間が経ったが、ついにやってみた。 何だこれバカ美味いんでやんの。 これ以外でもうコーラ飲みたくなくなるレベル。

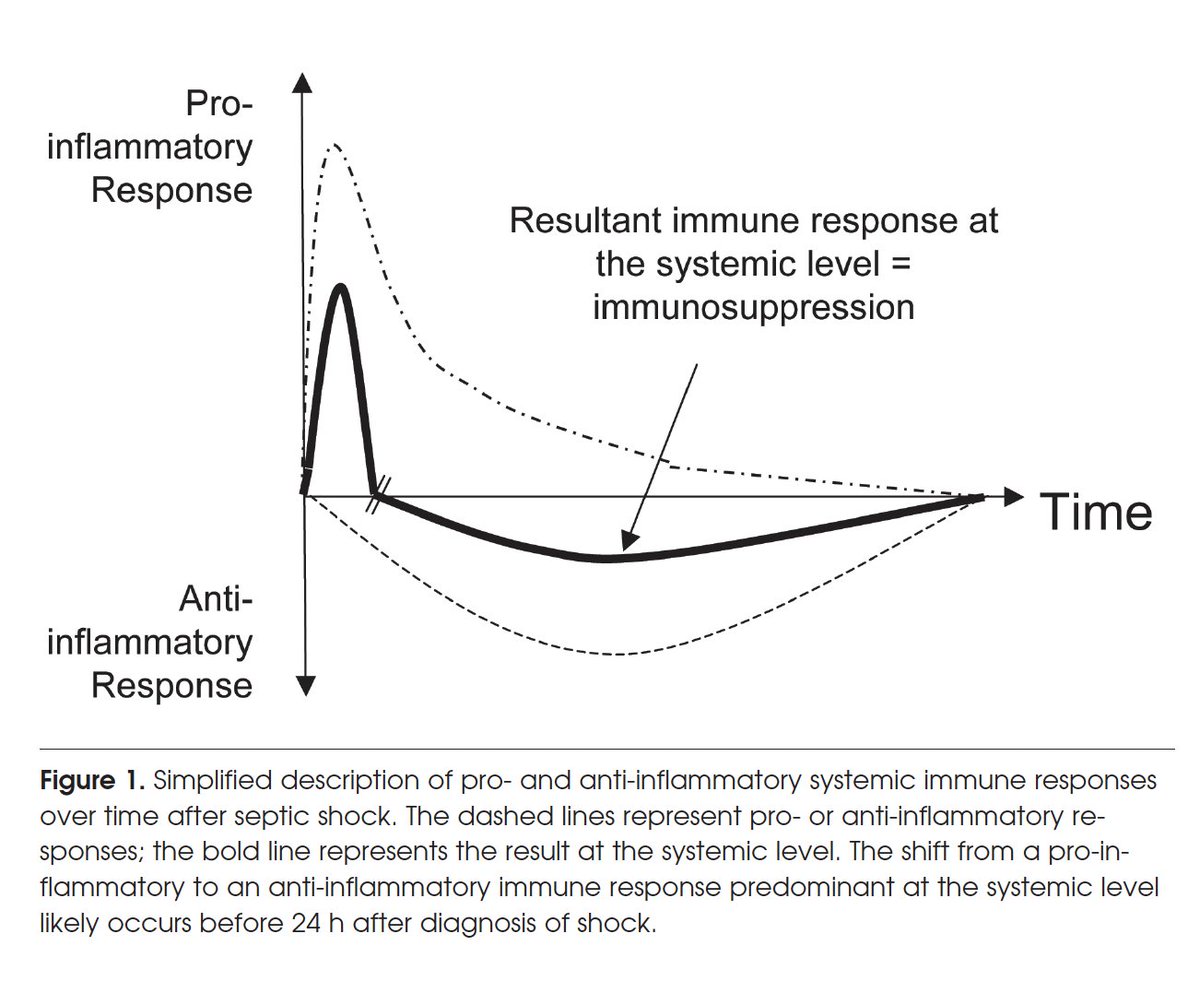
The more we learn about the power of IL-15 and the literature that exists decades ago (since 2010!) and never exploited is so exciting. Sepsis is responsible for over 200,000 deaths annually in the United States. We are planning a trial in community acquired pneumonia and sepsis. What is remarkable is the finding that Pseudomonas, a bacterial infection induces apoptosis (cell death) of NK cells, dendritic cells and CD8+ T cells contributing to mortality. More remarkable is the finding that IL-15 blocks this killing of these protective NK and CD8 T cells and improves survival. In this paper the final conclusion: "IL-15 prevents two immunopathologic hallmarks of sepsis, namely, apoptosis and immunosuppression, and improves survival in two different models of sepsis. IL-15 represents a potentially novel therapy of this highly lethal disorder." The more we learn, the more wondrous is this IL-15 cytokine molecule in our body! Our Sepsis Trial: ResQ218B-CAP clinicaltrials.gov/study/NCT07492… pubmed.ncbi.nlm.nih.gov/20026737/

Capital in place.Approvals in 34 countries. Indication expansion globally in motion. Pipeline of combination trials cooking. Immunotherapy 2.0. Never give up.


At ImmunityBio, we continue to prioritize long-term growth and the advancement of our broader immunotherapy platform. This additional non-dilutive financing supports our effort to expand globally with the goal of improving outcomes for patients. Read more: immunitybio.com/immunitybio-st…


@alc2022 @LoriMills4CA42 A very known surgeon from Dubai has told me that in Saudi Arabia they are using Anktiva for every cancer type. I expect lots of real world data coming soon. And it will be licensed OTC to ad on any cancer treatment and for lymphopenia.