

Jason Lee
552 posts

@JLeeLab
Assistant Professor @BCMhouston studying the logistics of subcellular organization. Membrane-bound and membrane-less organelle functions and contacts.









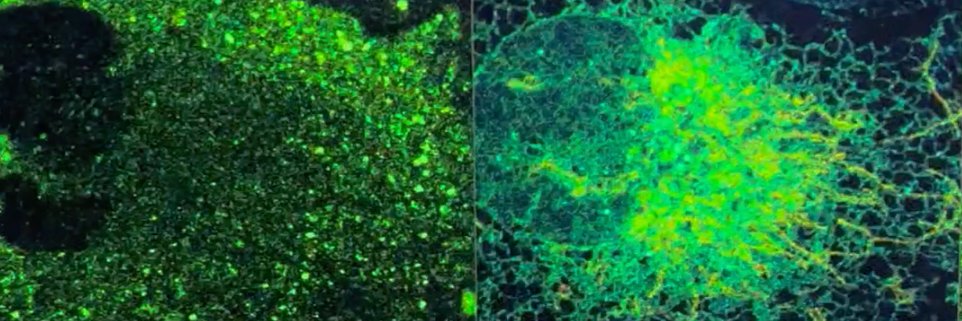
(10/10) Thank you to everyone in the @RibackLab and @Goodell_Lab, especially to @sciencyelmira, for this amazing work, and to @theNCI, @CPRITTexas, @LeukemiaRF, Searle Scholars, Ted Nash Long Life Foundation, and @BCM_MSTP for essential funding support.


Big news! Thrilled to welcome Dr. Kevin Klatt as our newest Assistant Professor in Nutritional Sciences! From Cornell → NIH → Baylor → UC Berkeley, he brings expertise in maternal & infant nutrition + micronutrients! @KCKlatt @DLOConnor0803 @uoftmedicine


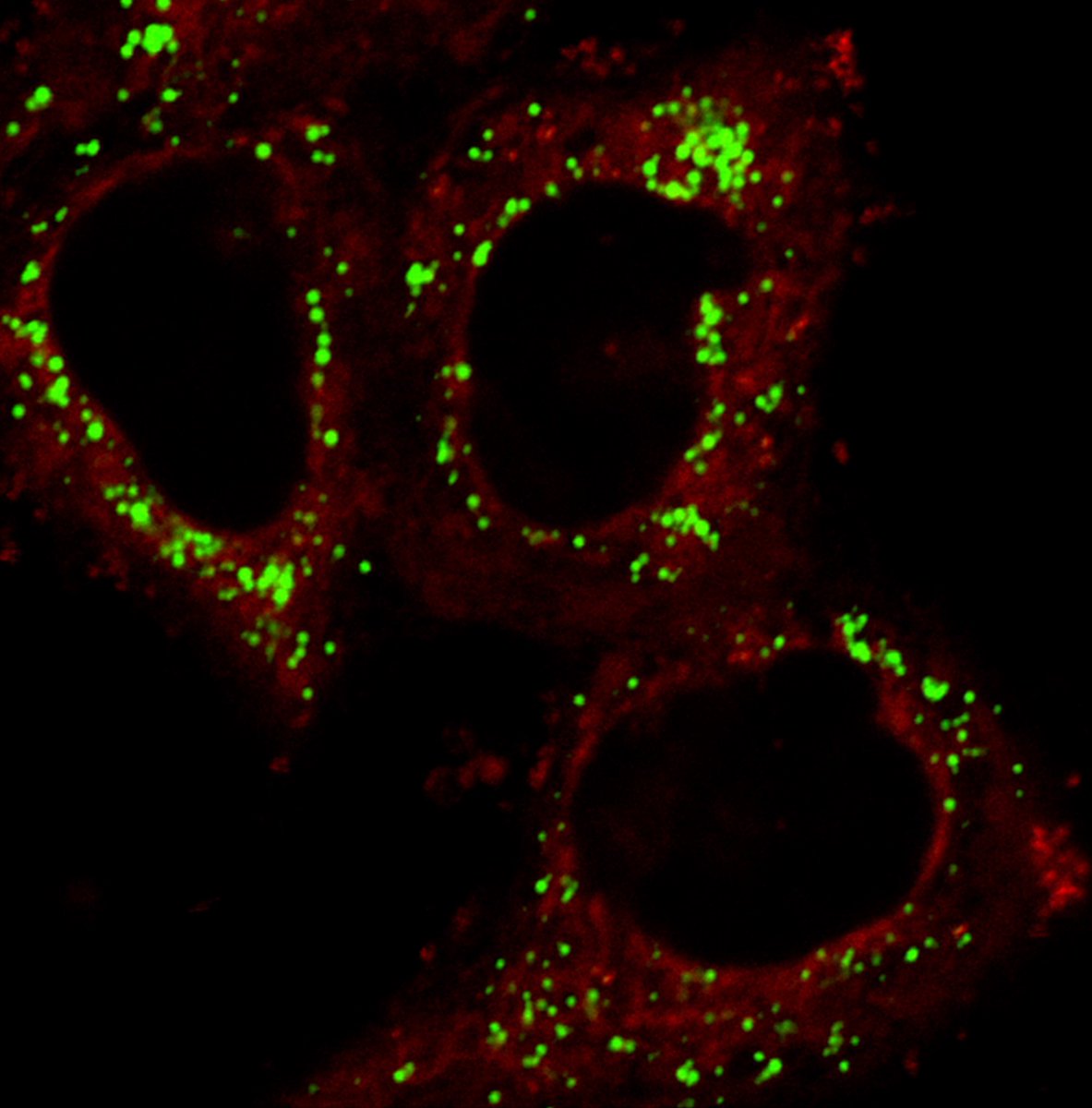



Excited to share our new Molecular Cell paper! We show that β-catenin is an adaptor that links canonical BAF (cBAF) with binding partners via IDR-domain interactions. (1/7) cell.com/molecular-cell…



