Houda Bahig, MD PhD
560 posts


@HoudaBahig
Radiation Oncologist & Clinician Scientist @chumontreal & @CRCHUM 🇨🇦 Views are my own

















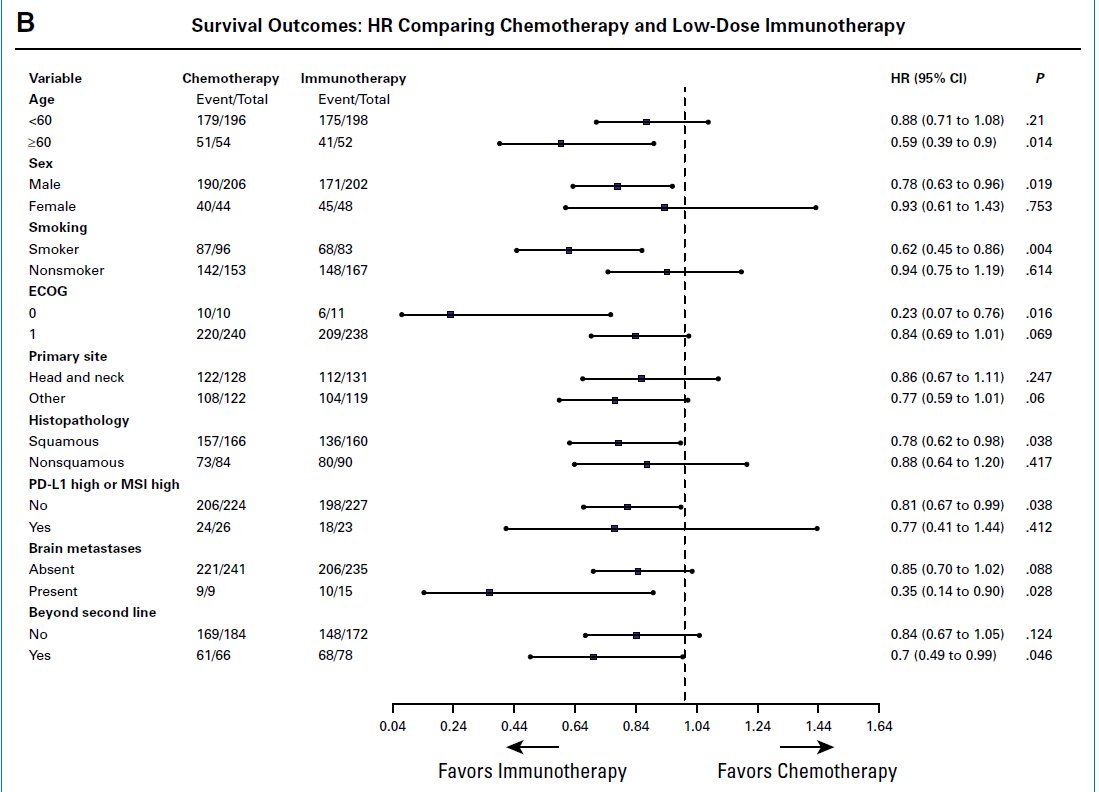






Question for those who’ve dug into KEYNOTE-689: If 43 pts had early ‘clinical progression’ (Table S2B), why isn’t there an early drop in EFS with neoadjuvant pembro? How do you reconcile this?



The abysmal state of global research funding on key domains such as surgery, radiotherapy and health services. Combined less than 4%. How have research funding organisations managed to get their strategic priorities so wrong? @cspramesh thelancet.com/journals/lanon…

Shocking! Only 1.4% of global cancer spend is used for surgical research and only 2.8% on radiotherapy research. Please help us learn how to cure more patients by funding curative therapies research! #radonc


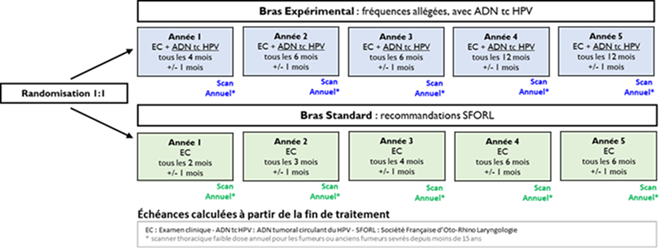
The linked paper is a nice document and summary product, but let's emphasize a few highlights: 1. Statement 16, strong consensus: ctHPVDNA should supplement conventional surveillance tools, rather than reducing or replacing them. Natural corollary: if it is not reducing or replacing surveillance, what is the point? 2. Statement 9, strong consensus: Detectable ctHPVDNA without clinical or radiographic evidence of disease often increases patient distress. 3. Statement 5, strong consensus: Large-scale, prospective randomized controlled trials are necessary to validate ctHPVDNA's role in HPV+ oropharyngeal cancer management. I would change that statement from validate to determine. Validate assumes it has a role today, but as the document states, it doesn't reduce or replace any currently established surveillance tools. There are so many exciting ways that ctHPVDNA may influence care (screening/diagnosis, de-escalation, non-imaging-based surveillance) but these strategies need to be established in prospective randomized trials.




The phase II randomized SUPPRESS-Lung trial evaluating SBRT for oligoprogressive NSCLC was closed today due to accrual challenges, before reaching its target. (NCT04405401) @drdavidpalma @DrAlexLouie @DrewMoghanaki @NormandBlais @_ShankarSiva @CJTsaiMDPhD @jefilion @CRCHUM