PeptiSense
132 posts




















Debt has consistently risen regardless of which party is in power











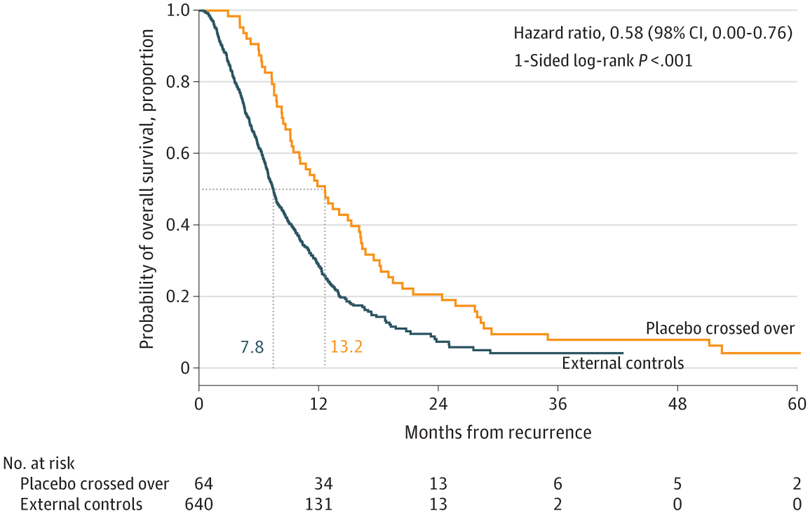
















$NWBO #DCVax Exciting news! @UCLAHealth has launched a clinical trial for a personalized cancer vaccine targeting aggressive brain tumors in adolescents and young adults. “Supported by the Department of Defense” 🟰#CancerMoonShot “This clinical trial represents a novel and potentially transformative approach to treating high-grade gliomas in children and young adults,” said Dr. Linda Liau. “We are optimistic that this research could lead to more advanced studies and eventually a new standard of care for this challenging subtype of brain cancer.” @ABTA @NBTStweets @IBTA @Braintumorfdn @braintumourrsch @BrainTumourOrg @ourbrainbank @HeadForTheCure @CancerCare @MacCancerSupport @MayoClinic uclahealth.org/news/release/u…