Prof. Dollar
6.6K posts


Prof. Dollar
@Prof_Dollar
Biotech enthusiast (evangelist) and long term investor. $QURE and $CLPT. Follow for updates on biotech, investing, stocks, entrepreneurship, greentech, and AI.

“In the US alone, about a thousand people die every year from Huntington’s, and if that’s avoidable, it seems to me that there is a responsibility on every nation’s government and regulator to do what they can to help the development of these drugs.”

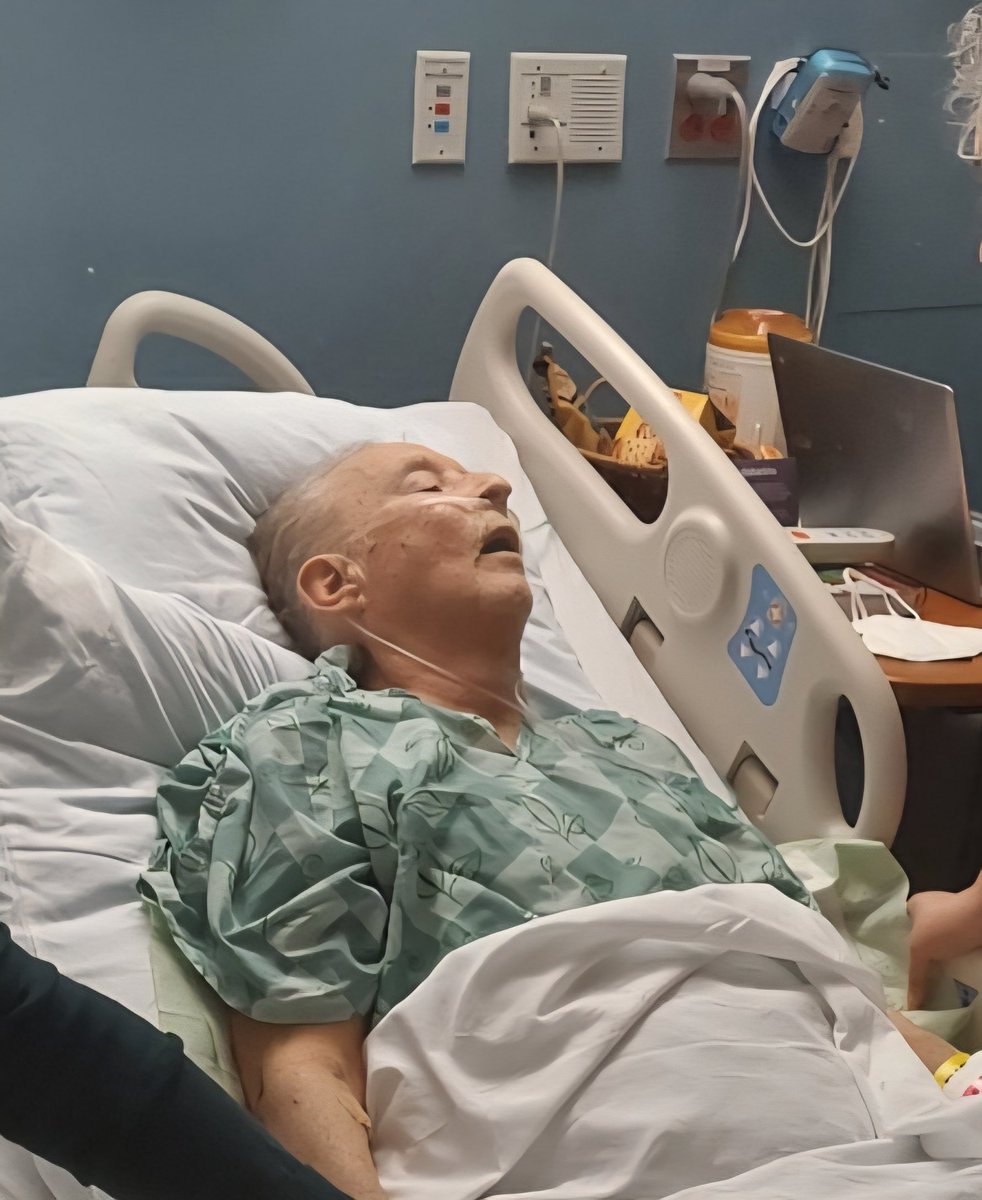
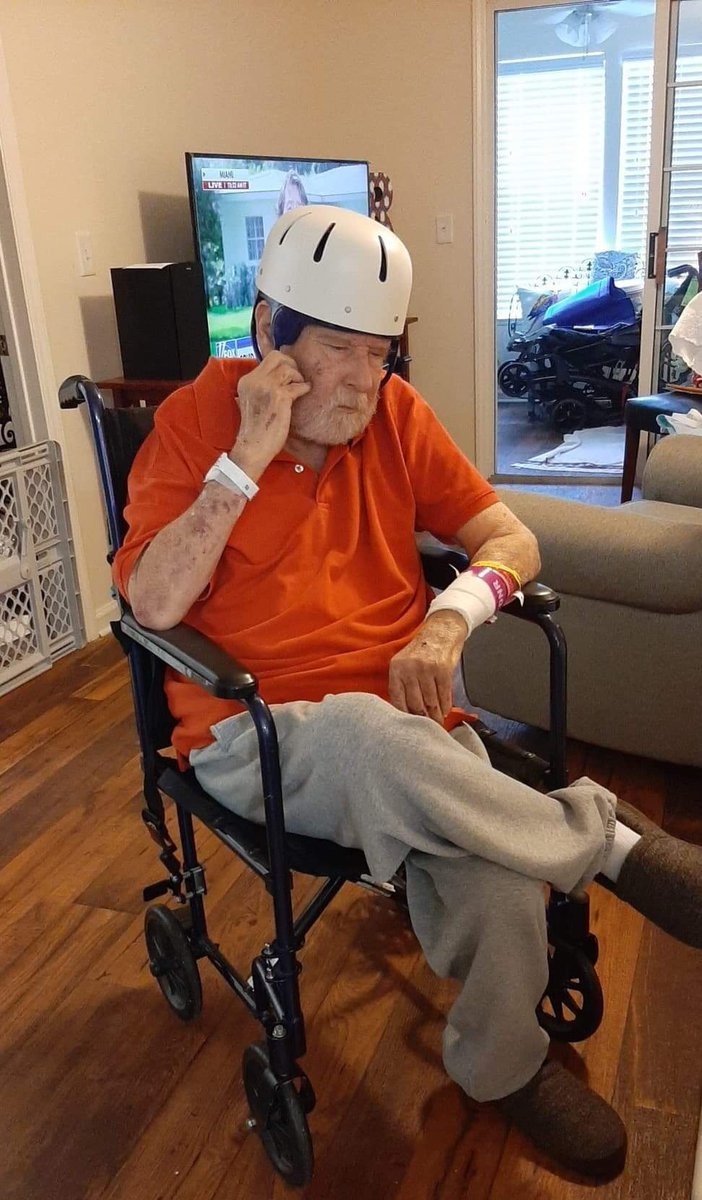

$qure $clpt Nice, more quotes from Ed Wild In a fireside chat at Advanced Therapies in London, Wild said he was concerned about the ethical implications of giving some participants a sham surgery despite the life-prolonging nature of AMT-130. “The agency stated there would need to be a sham arm for the entire duration of the clinical trial and treatment arm,” Wild told BioXconomy. “Patients would have to go 3–5 years without treatment, which is a very long time, to be honest. A substantial portion may advance so much [in their disease progression] that they are no longer eligible for treatment.” Wild admits that he and his colleagues were blindsided in the sudden U-turn the agency took after the recent US election, stating it vastly contradicted the agency’s previous position. “We were surprised that the agency took such a substantial step away from the advice it had previously given,” Wild elaborated. “It introduces an air of unpredictably that may be by design, but it makes life very difficult for patients.” “It’s very difficult to see how it becomes ethical to enroll people in a surgical trial, knowing that inevitably some would be too advanced to receive their treatment.” “All I can hope is that the combination of the patient voice, which is loud and clear, and the current data we have, and the emerging data will be heard,” stated Wild. “I hope the agency keeps an open mind and is receptive to what can and can’t be achieved, both ethically and practically.” “In the US alone, about a thousand people die every year from Huntington’s, and if that’s avoidable, it seems to me that there is a responsibility on every nation’s government and regulator to do what they can to help the development of these drugs.” @DesertDweller93 @Prof_Dollar @laurencurehd bioxconomy.com/modalities/fda…

$qure $clpt Nice, more quotes from Ed Wild In a fireside chat at Advanced Therapies in London, Wild said he was concerned about the ethical implications of giving some participants a sham surgery despite the life-prolonging nature of AMT-130. “The agency stated there would need to be a sham arm for the entire duration of the clinical trial and treatment arm,” Wild told BioXconomy. “Patients would have to go 3–5 years without treatment, which is a very long time, to be honest. A substantial portion may advance so much [in their disease progression] that they are no longer eligible for treatment.” Wild admits that he and his colleagues were blindsided in the sudden U-turn the agency took after the recent US election, stating it vastly contradicted the agency’s previous position. “We were surprised that the agency took such a substantial step away from the advice it had previously given,” Wild elaborated. “It introduces an air of unpredictably that may be by design, but it makes life very difficult for patients.” “It’s very difficult to see how it becomes ethical to enroll people in a surgical trial, knowing that inevitably some would be too advanced to receive their treatment.” “All I can hope is that the combination of the patient voice, which is loud and clear, and the current data we have, and the emerging data will be heard,” stated Wild. “I hope the agency keeps an open mind and is receptive to what can and can’t be achieved, both ethically and practically.” “In the US alone, about a thousand people die every year from Huntington’s, and if that’s avoidable, it seems to me that there is a responsibility on every nation’s government and regulator to do what they can to help the development of these drugs.” @DesertDweller93 @Prof_Dollar @laurencurehd bioxconomy.com/modalities/fda…


Everyone following the @uniQure_NV $QURE AMT130 saga needs to read and retweet this. The rot in the @US_FDA begins at the top and needs to be fixed NOW! @SenRonJohnson @SenRickScott @RepAuchincloss @adamfeuerstein @l_e_whyte @laurencurehd @BeckyQuick turn up the heat.

CSL's #hemophilia B gene therapy, licensed from $qure, is slowly becoming a success story, no matter the temporary manufacturing setback: $57M in H2 2025 sales following a slow start in 2023/4, and 51 out of 54 patients prophy free after 5 years! $sgmo

If a drug is already widely used in humans abroad, the FDA shouldn’t require routine testing in animals.


30,000 Americans are symptomatic with Huntington's disease right now. Another 200,000 carry the gene. There is no approved treatment that slows progression. None. AMT-130 showed 75% slowing at 36 months. The 48-month data drops this summer. If the numbers hold, the FDA will have to explain why it spent months calling this therapy "failed" instead of reviewing it. $QURE #HuntingtonsDisease #AMT130

Another impressive result for the national priority voucher program. 54 days for a full FDA review! 🇺🇸

For those who don’t have access to the FT. Below is the article on Arnold Ventures from December directly linking Vinay and Katherine and specifically calling out uniqure $qure @DesertDweller93 @biggercapital @peter_mantas @MartinShkreli @canoebrookbl @adamfeuerstein

For those who don’t have access to the FT. Below is the article on Arnold Ventures from December directly linking Vinay and Katherine and specifically calling out uniqure $qure @DesertDweller93 @biggercapital @peter_mantas @MartinShkreli @canoebrookbl @adamfeuerstein

caveat here is that it was routine when Prasad ran CBER (at least 4 overrules in Prasad's 10 mo. tenure) but it didn't/doesn't still seem to be happening elsewhere in FDA



Pharma has optimized research portfolios for ease of FDA approval & pricing power rather than on drugs most valuable for society. It's why we @Arnold_Ventures work to improve how drugs are approved & priced to deliver better health outcomes at lower costs. atelfo.github.io/2023/12/23/bio…




