Ross Keller, PhD
986 posts


@RossK11
Senior Research Director at Private Health Management. Previously, Fellow @sloan_kettering and PhD studying cancer evolution @Penn_State. Posts my own.





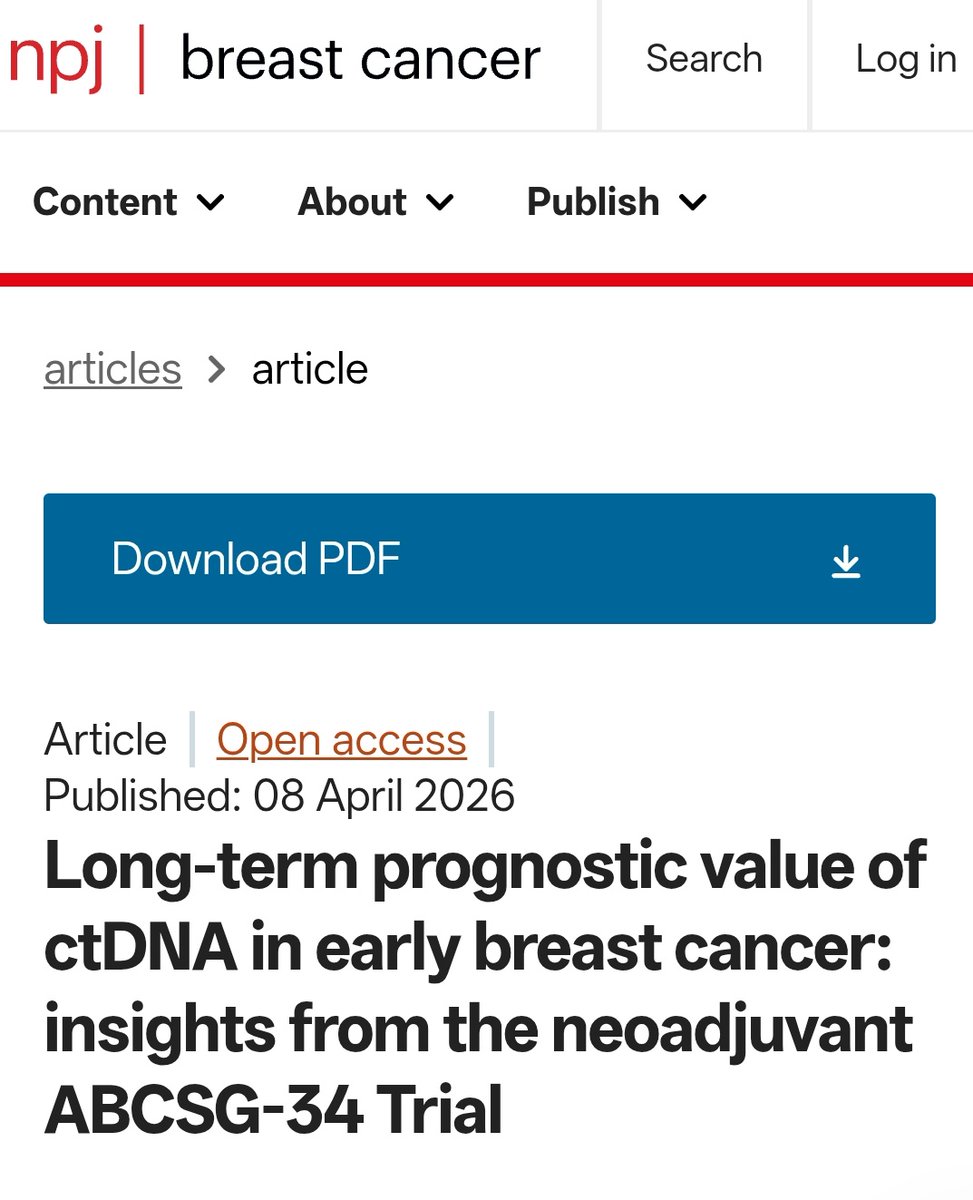







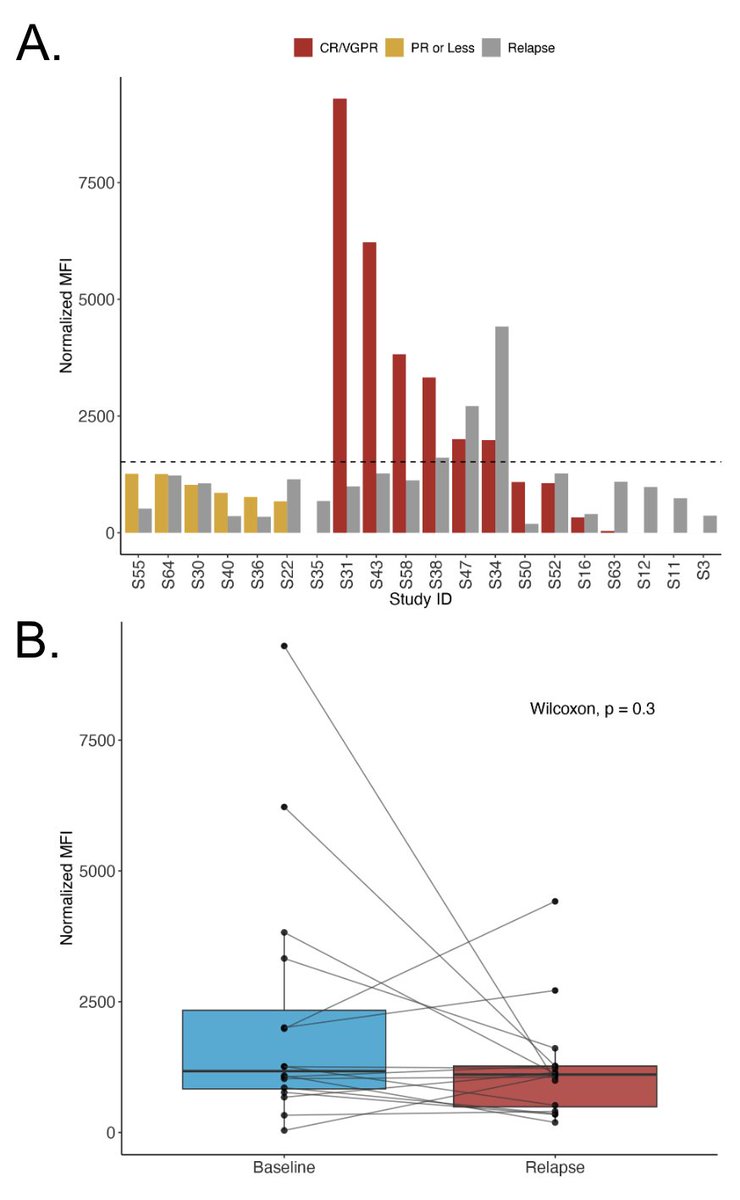




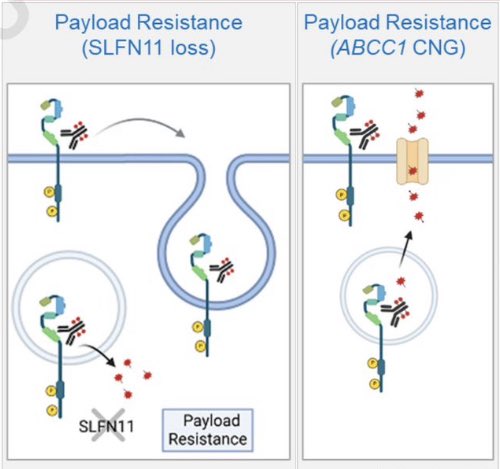
Do HER2 mutant #NSCLC tumors progressing on T DXd lose HER2 dependence? No. Resistance is mainly driven by payload related mechanisms such as SLFN11 loss or ABCC1 gain, or impaired antibody binding due to domain IV mutations, rather than loss of the HER2 driver. HER2 signaling persists, and tumors retain sensitivity to #HER2 TKIs (zongertinib/poziotinib). Progression on #TDXd opens a rational path to TKIs jto.org/article/S1556-… @OncoAlert @AndresFCardonaZ

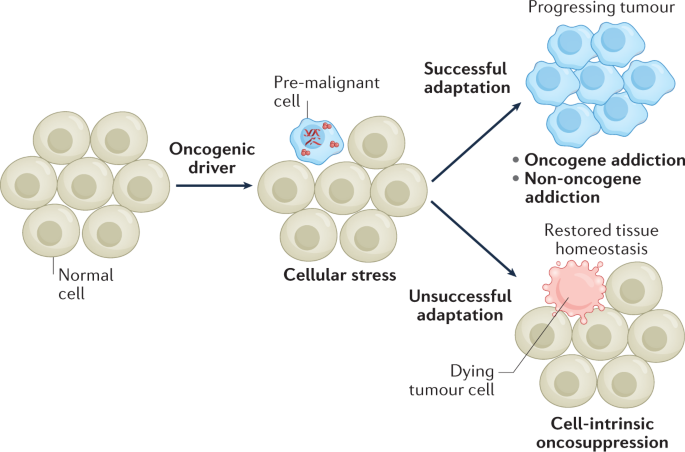

Cancer is often described as a genetic disease—but that statement is incomplete unless we understand which mutations actually matter. Tumours accumulate thousands of genomic alterations over time. However, not all of them contribute to the disease. This distinction led to one of the most important conceptual frameworks in oncology: driver versus passenger mutations. ✳️ Driver mutations are the ones that change the rules of the game. They confer a selective advantage to the cell, allowing it to proliferate faster, evade apoptosis, adapt to hypoxia, or escape immune surveillance. In evolutionary terms, they are under positive selection. Without them, cancer does not develop. At the molecular level, drivers typically affect three major classes of genes. - Oncogenes become constitutively active (for example, mutations in KRAS or BRAF). - Tumour suppressor genes are inactivated (such as TP53). - DNA repair systems are compromised (e.g. BRCA1). The result is not just growth, but uncontrolled, deregulated growth with evolutionary potential. Importantly, not all drivers appear at the same time. Some occur early and are present in all tumour cells (so-called truncal drivers), while others arise later in subclones, contributing to intra-tumour heterogeneity and therapeutic resistance. ✳️ In contrast, passenger mutations are biologically neutral—at least in principle. They accumulate as a by-product of genomic instability, particularly in tumours with defective DNA repair. These mutations are not under selection and do not directly contribute to the malignant phenotype. In many cancers, the vast majority of detected mutations fall into this category. However, dismissing passengers as irrelevant would be an oversimplification. - First, they provide a molecular record of the tumour’s evolutionary history, allowing reconstruction of clonal dynamics. - Second, some passenger mutations generate neoantigens, which can be recognised by the immune system—this is one of the mechanistic bases underlying the association between high mutational burden and response to immunotherapy. The key challenge, both experimentally and clinically, is therefore not detecting mutations—but distinguishing signal from noise. In essence, cancer genomes are not just chaotic. They are structured by selection, where a small number of driver events shape the disease, and a vast background of passengers reflects the instability that fuels it.



We thought KRAS was the engine So it was turned off But tumors adapt, rerouting the pathway In this preclinical study in KRASm NSCLC, loss of PTPRR activates EGFR, reactivating ERK and driving resistance to sotorasib/adagrasib. KRAS is blocked EGFR turns the pathway back on That’s why vertical blockade (KRAS + EGFR) may matter, remember that preclinical study 🇪🇸 ❓ Will panRAS inhibitors be enough? Preclinical data suggest probably not. Combos may be the answer aacrjournals.org/cancerrescommu… @OncoAlert @Aiims1742