Stephen Wang
6.1K posts



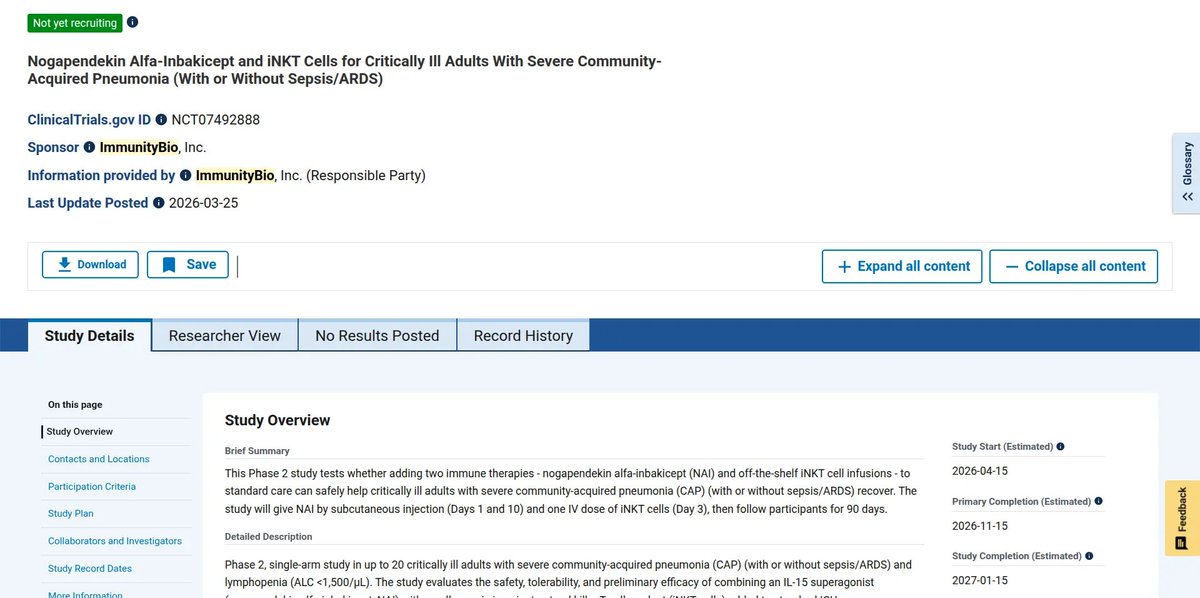
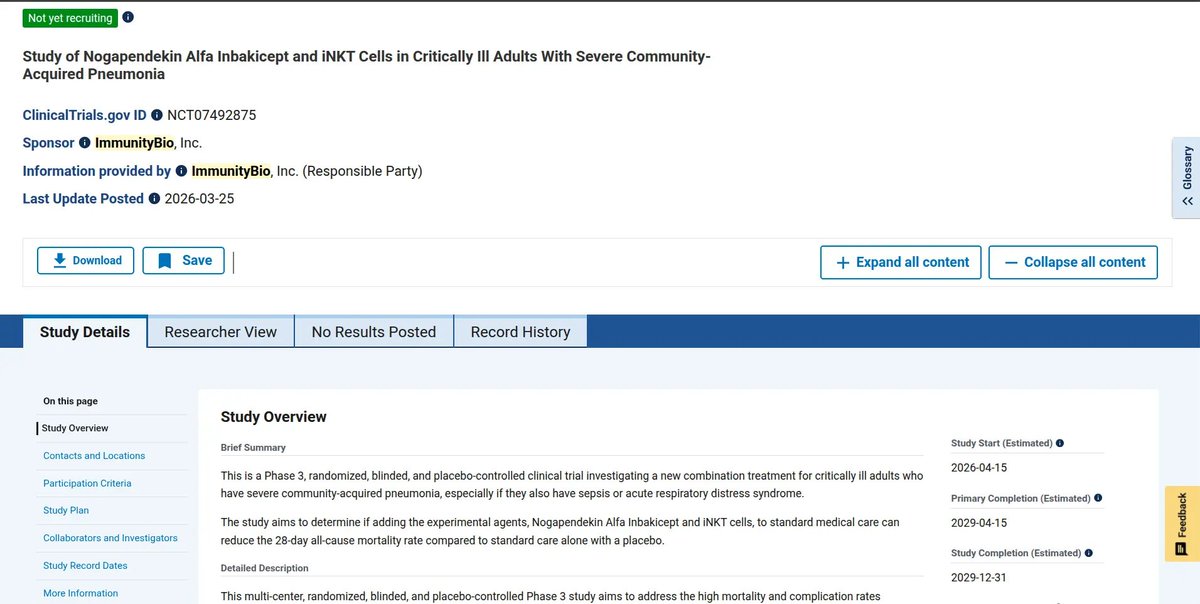
$IBRX new Phase 1 Pancreatic TRial listed with Anktiva
















TERAFAB: the next step to becoming a galactic civilization Together with @SpaceX & @xAI, we're building the largest chip manufacturing facility ever (1TW/year) – combining logic, memory & advanced packaging under one roof. To harness as much power as possible from the Sun, we need to send 100 million tons of solar capture into space – per year. This requires massive scale. – Capability to launch millions of tons of mass into orbit – Solar-powered AI satellites – Millions of @Tesla_Optimus robots to help build it out All of these need chips: 100-200GW of chips for Optimus alone, plus terawatts for solar-powered AI satellites. That's more than all the chip manufacturers in the world combined can provide today, or even by 2030 (based on projected production growth). We're building TERAFAB to close the gap between today’s chip production & the future's demand – a future among the stars terafab.ai





🚨 “If Iran doesn’t FULLY OPEN, WITHOUT THREAT, the Strait of Hormuz, within 48 HOURS from this exact point in time, the United States of America will hit and obliterate their various POWER PLANTS, STARTING WITH THE BIGGEST ONE FIRST…” - President DONALD J. TRUMP


🚨🇺🇸🇮🇷 BREAKING: Trump gives Iran a 48-hour ultimatum to fully open the Strait of Hormuz or the U.S. will obliterate their power plants, "starting with the biggest one first." Three weeks of "winding down" talk just ended with the most aggressive threat of the entire war. Destroying Iran's power grid would plunge 90 million people into darkness and trigger a humanitarian catastrophe. The next two days will define everything. Source: Truth Social











