Michael Shusterman, MD
1.4K posts


Michael Shusterman, MD
@guildsman
Gastrointestinal Medical Oncologist @Perlmutter_CC, Associate Program Director @nyulisom_HemOnc. Tweets my own. #Meded







New in #practicalRO: Adaptive Salvage Stereotactic Radiation for Locoregional Recurrence of Pancreatic Adenocarcinoma After Surgery. #radonc tinyurl.com/prolakomy

The AGITG CONTROL NETS is finally out in paper! Does the CAPTEM-PRRT combo perform better than CAPTEM in patients with pancreatic NETs? Well, there is more in this paper than that but let's focus on the pNETs. Bottom line: Complex trial, many moving parts, small cohort, does not and should not change practice. More thoughts: The study would suggest so with longer PFS in the PRRT/CAPTEM but the bigger question is how patients who got PRRT alone compare but that was not done here. Keep also in mind that the number of treated patients was very small. The PFS in the PRRT/CAPTEM group was 59.4 months which is very impressive (For PRRT alone reference, it was 20.7 months in OCLURANDOM and 24.5 months in COMPETE). There were only 2 patients (3%) among those who had PRRT who developed therapy-related myeloid dysplasia which is reassuring but also goes against what others have reported where the risk is as high as 10%. Will this change anything for me? Absolutely not as I would need larger studies to be convinced and also, I am not sure the therapy sequencing matters that much as long as you get all the effective therapy during the illness. But what this study does is to help revive the concept of chemo-PRRT and support the development of other trials looking at combining PRRT with systemic therapy and @AmanChauhanMD, I am looking at you now... 😉 sciencedirect.com/science/articl…

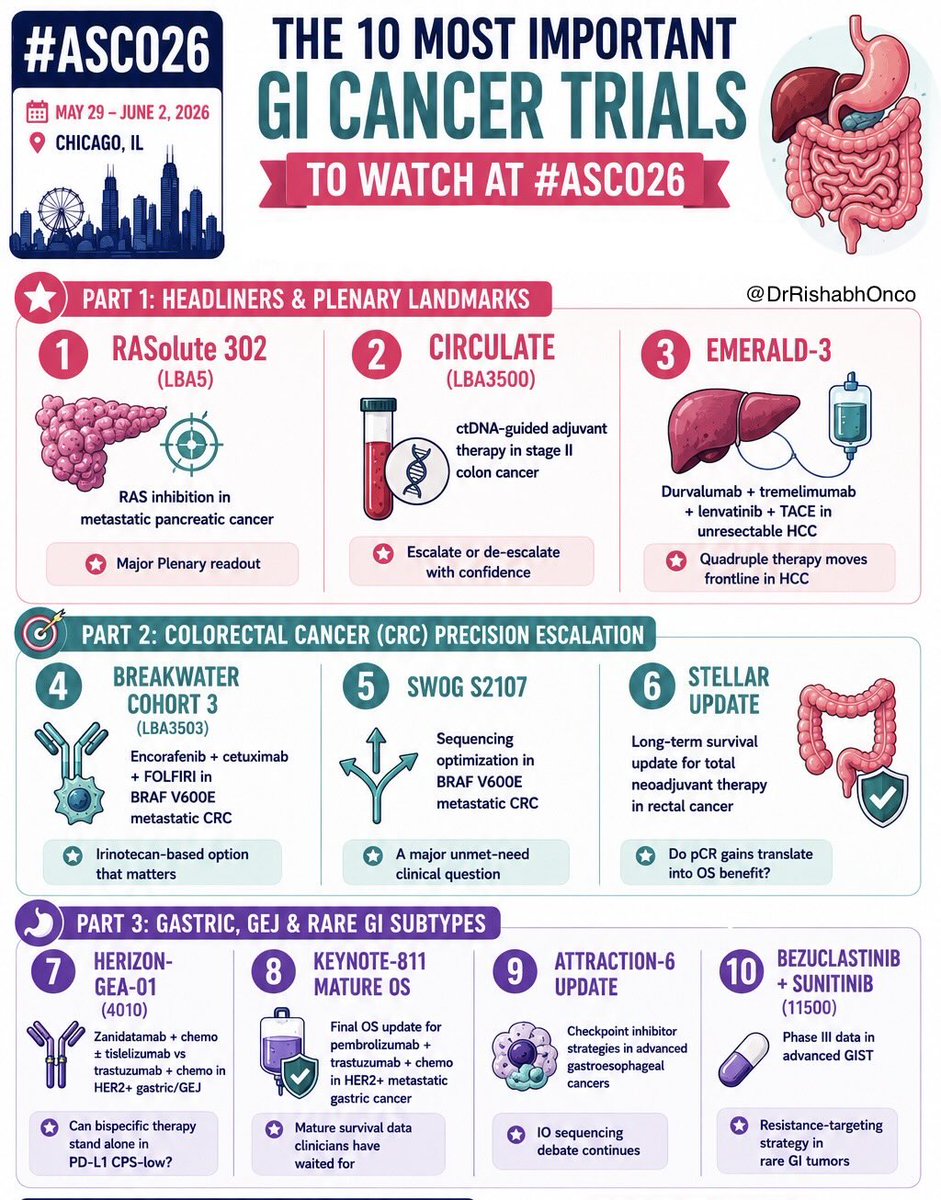
🚨 THE 15 MOST IMPORTANT TRIALS OF #ASCO26 May 29 - June 2 | Chicago Which trial are you watching most closely? 🌟 PLENARY GAME-CHANGERS 1️⃣ PROTEUS Perioperative apalutamide + ADT in high-risk localized prostate cancer 2️⃣ LIBRETTO-432 Adjuvant selpercatinib in RET+ NSCLC 3️⃣ HARMONi-6 Ivonescimab + chemo vs tislelizumab + chemo in squamous NSCLC 4️⃣ RASolute 302 Daraxonrasib (RMC-6236) in metastatic pancreatic cancer 5️⃣ SARC041 Abemaciclib in dedifferentiated liposarcoma ⚡ FRONTLINE & PERIOPERATIVE SHIFTS 6️⃣ KEYNOTE-B15 / EV-304 EV + pembrolizumab vs chemo in MIBC 7️⃣ LITESPARK-022 Pembrolizumab + belzutifan in adjuvant ccRCC 8️⃣ AMBITION Paclitaxel/bevacizumab ± atezolizumab in HR+ breast cancer 9️⃣ NeoADAURA Neoadjuvant osimertinib in EGFR+ NSCLC 🔟 A-DREAM ADT interruption strategies in mCSPC 🧬 PRECISION, ADCs & NEXT-GEN IMMUNOLOGY 1️⃣1️⃣ DESTINY-Breast06 T-DXd expands into HER2-ultralow disease 1️⃣2️⃣ CROWN (7-year update) Lorlatinib durability in ALK+ NSCLC 1️⃣3️⃣ DeLLphi-312 Tarlatamab in frontline SCLC 1️⃣4️⃣ COMMIT Atezolizumab + FOLFOX/Bev in MSI-H mCRC 1️⃣5️⃣ IMvigor011 ctDNA-guided adjuvant atezolizumab in bladder cancer #OncoTwitter #MedTwitter #ASCO26 #CancerResearch @OncoAlert @ASCO @JCOPO_ASCO @OncBrothers







Completion of all planned perioperative therapy was associated with best outcomes in MATTERHORN 🗻🗻. Intuitive but an important signal when counseling patients- dose reduce if you must but try to complete. @GIMedOnc @UGrewalMD @TheGutOncLab asco.org/abstracts-pres…





Managing toxicity is hard. It is part science, part judgment, and part art. And it is not always fully spelled out in the package insert. In advanced GI cancers, we spend a lot of time talking about the next drug, the next target, the next trial. But for many patients, the most important question is more practical: Can we deliver effective therapy in a way they can actually tolerate? Dose modifications. Schedule adjustments. Quality of life. This is the real world of oncology. Grateful to publish this review with @GutOncLab, @UGrewalMD @TimothyJBrownMD @guildsman on optimizing systemic therapy for advanced GI cancers. Personalized dosing is not “less aggressive” care. Done thoughtfully, it is often better oncology. @OncoAlert @Onco_Nexus clinical-colorectal-cancer.com/article/S1533-…

Invited commentary on: “Post-surgical ctDNA as a prognostic biomarker for relapse of resected pancreatic ductal adenocarcinoma” sciencedirect.com/science/articl…









Phase 1/2 data for Daraxonrasib are now published in @NEJM - Patients with pretreated PDAC harboring KRAS, NRAS, or HRAS mutations at codons 12, 13, or 61- received Daraxonrasib once daily in 21-day cycles at doses ranging from 10 mg to 400 mg. MTD was not reached, 400 mg was maximum administered dose. 🚨📛Important to highlight the toxicities here (300 mg, n=83)- rash (in 90%), stomatitis or mucositis (in 54%), diarrhea (in 52%), and nausea (in 39%)--Grade 3 or higher occurred in 34% of the patients. Dose modifications:48% (dose interruption: 43% and dose reduction: 30%). However, no patients discontinued treatment due to AEs. 📈📊Now looking at efficacy- at a median follow-up of 17 months, mPFS=8.5 months, mOS=13.1 months. ORR 29% with median time until objective response= 2.6 months and duration of response= 8.2 months. 👏🔥 💯💯Overall impressive data for efficacy but it is obvious that this comes at a price, i.e., toxicity! But this is true for any drug, especially TKIs- rash, GI tox, etc are not new toxicities with TKIs. I bet as our experience with these agents grows, we will find ways to minimize these and preserve QoL as our patients continue to benefit from these game changing novel agents. This sets the stage for whats coming at #ASCO26 nejm.org/doi/full/10.10…

