Nathan Lew
1.4K posts

Nathan Lew
@lewdubbs
Long: Injectables + Short: Injections // Former founder & CEO @ https://t.co/AKOhSTBzY3 (acquired) // advisor + investor.
Santa Monica, CA Katılım Aralık 2008
1.3K Takip Edilen486 Takipçiler
Nathan Lew retweetledi

The first-ever, peer-reviewed expert consensus on the use of ketogenic metabolic therapy for serious mental illness.
This field is taking off!
frontiersin.org/journals/nutri…
English

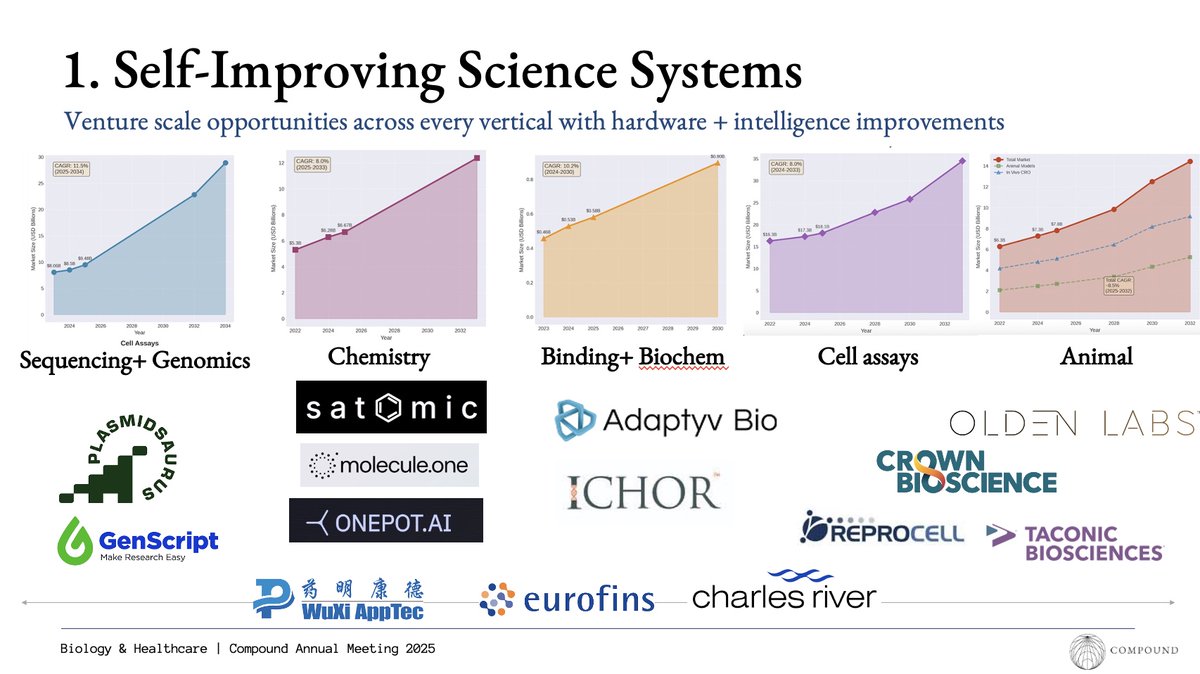
a friend asked me recently about the business of selling new hardware to biotech companies and whether this was a venture scale business.
from studying the market, my takeaway from new hardware is that it requires many companies to change their current hardware they spent 6-7 figures on already. not only this but the sales are hard bc automation people aren't that senior so you get stuck between automation people and c-suite in sales process. this doesn't mention the one-off vs. recurring payments you get in other lab adjacent businesses such as software or repeatable services.
this is why I'm excited about companies using automation to make step change CROs. put another way selling services not hardware. an example is plasmidsaurus and think similar companies can be built on different layers of chemistry, therapeutic assays, and animal testing. on the bigger company side, Ginkgo and Emerald also seem to have a general experimental offering that's exciting to see and has some solid traction to boot.
in a way, I see these centralizing experimental workflows but still thinking about centralized experimentation vs. decentralized trials.
English

@DrCamRx I think that’s (unintentionally) happening to some degree with FMT
English
Nathan Lew retweetledi
Nathan Lew retweetledi

Overthinking is imagination trapped in prediction.
When you think too much, your brain’s Default Mode Network takes over.
It loops on “what ifs,” “should I,” “what could go wrong.”
This network was meant for creativity, but when hijacked, it becomes simulation instead of creation.
You start rehearsing life instead of living it.
Action doesn’t come from thinking harder.
It comes from shifting networks.
The moment you move—walk, speak, draw, breathe with intention—the brain transitions from the predictive DMN to the salience and sensorimotor networks.
These systems don’t analyze; they engage.
Neurons stop firing in circular patterns and start firing in directional patterns. Your body STOPS ruminating and enters into real-world experiences.
English
Nathan Lew retweetledi

Do you have a Return Home to Family Protocol? Because DJ Shipley does & it’s about regimen and presence for you so the others in your life can be more spontaneous and feel more support. Impressive. Check it out.
Andrew D. Huberman, Ph.D.@hubermanlab
This is how DJ Shipley returns home each day to his wife and kids. It’s as regimented as his morning routine- nothing left to chance (on his side). Anyone trying to balance work and personal life should listen to this.
English

@HansAmato Or donate blood once a quarter, no? Doesn’t that lower ferritin?
English

Too much iron ages you faster.
Aspirin might be your secret weapon.
Aspirin lowers iron...not by causing bleeding, but by turning into iron-binding compounds.
Up to 20% of elderly on low-dose aspirin (75–100mg/day) develop iron deficiency anemia without GI bleeding.
Here’s why:
Aspirin is metabolized into salicylic acid, salicyluric acid, and dihydroxybenzoic acids, all of which are potent iron chelators.
These metabolites mobilize iron from tissues and bind it, leading to gradual but significant iron loss through excretion.
Iron excess kills you slowly. Lowering it is beneficial.
English
Nathan Lew retweetledi
Nathan Lew retweetledi
Nathan Lew retweetledi
Nathan Lew retweetledi

We are moving the state of our incorporation from Delaware to Nevada.
It used to be a no-brainer to start a company and incorporate in Delaware. That’s no longer the case.
We're not the first - Dropbox, Tripadvisor and Tesla have already left.
Why we're moving (and why you should consider too):
- Business Judgment Rule: Delaware’s rule is based on judicial decisions and can change. Nevada codifies it in statute, making it more predictable and less subject to court reinterpretation.
- Liability: Nevada protects directors and officers from personal liability unless a plaintiff can prove intentional misconduct, fraud, or knowing legal violations.
- Books + Records: In Nevada, only shareholders with 15% or more ownership can inspect records, reducing legal fishing expeditions.
- Courts: Delaware’s court advantage is fading. Nevada is modernizing with judge appointments and jury trial waivers to handle complex business disputes.

English
Nathan Lew retweetledi
Nathan Lew retweetledi

Aim for 7-8 hours
Still kick ass when get less than 4
Health and resilience dichotomy
if you can't function in un-ideal circumstances, you are not robust and healthy
CoffeeBlackMD@CoffeeBlackMD
We have now moved into a ridiculous over-prioritization of sleep. And most of you have gone full neurotic. Some completely paralyzed by it.
English
Nathan Lew retweetledi

Creatine is not just for your muscles, it is for your brain.
Researchers gave people with Alzheimer's creatine, and it raised the level of creatine in the brain, which had a positive effect on their memory.
Personally, I've seen cognitive benefits dosing at 15 grams or more.
More studies need to be done, but this is promising.

English
Nathan Lew retweetledi

The Truth about BPC-157: Why it isn’t FDA Approved and Why It Will Never Be
BPC-157 is one of the hottest compounds on the market right now with many people, especially in the bodybuilding and functional medicine space swearing by its beneficial effects for gut health and joint healing, among a whole host of other proposed possible benefits.
It is not however, a FDA approved pharmaceutical. Clinicians are prescribing it through compounding pharmacies without a clear, approved indication and people are buying it as a “research chemical” for “not human consumption” online. After ending up on the FDA “do not compound” list in late 2023, sources of injectable BPC have become scarce. Some have relabeled it as Pentadecapeptide Aringate or PDA (changing the acetate to an arginate) and are prescribing it for subcutaneous administration that way, while the confusion about its status has led to a vibrant oral capsule market for BPC in what appears to be a grey area of legality. (did the FDA accidentally make this into an OTC supplement by adding it to the do not compound list?)
So if it's so effective, why is it not approved by the FDA for human use? Is there some sort of conspiracy against “good” medicines that have potential to “heal” by big pharma? Are they worried it may cut into the profits of their other drugs, such as NSAIDs, PPIs, and biologics?
While those who stand to profit from BPC-157 sales on the internet would have you believe that, the answer is no.
As far as I can tell in my deep inquiry there is no global conspiracy against BPC-157. There are, however, simple economics of drug development processes that make it so BPC-157 likely will never go through the full process of drug approval because of financial incentives.
I would even venture to believe that a pharmaceutical company like Merck would LOVE to have the exclusive rights to BPC-157. However, because of the patents being filed and the way patents work on small peptides, pushing BPC through the entire FDA product, which would cost millions of dollars, is not going to happen.
Let’s look at how we got here:
BPC-157 is not new. Croatian researchers isolated the full, larger peptide BPC in 1998 from gastric juices and found the 15 amino acid sequence, BPC-157 to be the active part responsible for its clinical effects (the story of how it was discovered is fascinating, but that is a conversation for a different day if there is enough interest, let me know below). The first human safety study on health volunteers was conducted in 2002 and a “phase 2” double blind, randomized clinical trial (small and not sufficient for US phase 2 purposes) was conducted in 2005 with BPC-157 enemas for ulcerative colitis, with some promising effects (see research name PL 14736 to find the references). Yes, there were human studies with BPC-157 showing no known adverse effects and safety, however since those two initial trials it appears almost all the data has been in animal models, with a few, small studies here and there for human use.
If only 10% of the animal data on BPC-157 ends up being valid and applicable to humans, this is a blockbuster drug/compound that has great potential for human use. But this also may be part of the problem of pushing BPC through regulatory constraints (yes BPC-57 is suffering from success).
To get a drug FDA approved, a company must conduct phase 1, 2 and 3 clinical trials proving safety and efficacy of the compound through a laborious, multi year and extremely costly process. We’re talking a decade or more and hundreds of millions of dollars. The drug needs to be proven to work for a specific disease indication to get approval, while BPCs effects seem to be pleiotropic and the research cannot seem to focus on one, singular disease case to focus on.
Diagen is the company that currently owns the BPC-157 US patent (the European patent is expired), which was filed in 2012, approved in 2017 with an expected expiration of 2033. These are all the conditions listed in the patent:
Conditions and Disorders:
1. Stress-Related Diseases & Disorders
2. Gastrointestinal Conditions
• Ulcers in any part of the gastrointestinal tract
• General anti-inflammatory activity
• Gastrointestinal inflammatory diseases
• Crohn’s disease
• Acute pancreatitis
3. Organoprotective Needs
• Diseases, conditions, or disorders requiring organoprotective activity
4. Viral Infections
• Hepatitis A
• Herpes strains
• Influenza A
• Arthropod-borne (ARBO) viruses, including:
• Tick-borne encephalitis
• West Nile fever
• Dengue (types 1-4)
• Cytomegalovirus (CMV)
• Lymphocytic choriomeningitis (LCM) virus
• Feline leukemia virus
5. Oncology & Tumors
• Melanoma and related tumors
6. Tissue & Wound Healing
• Accelerated healing of wounds, burns, and bone fractures
• Regeneration of ruptured nerve linkages
• Achilles tendon and muscle repair
• Spinal cord injury recovery
7. Nitric Oxide (NO)-Related Disorders
• Hypertension
• Hypotension
• Anaphylaxis
• Circulatory and septic shock
• Thrombocyte aggregation
8. Neurological & Autoimmune Disorders
• Multiple sclerosis
• Myasthenia gravis
• Lupus erythematosus
• Neuropathy
• Somatosensory nerve dysfunction
• Asthma
• Rhinitis
• Pemphigus
• Eczema
9. Neurochemical & Substance-Related Disorders
• Catecholaminergic dysfunction
• Schizophrenia
• Withdrawal effects from amphetamines, drugs, and alcohol
10. Corticosteroid & NSAID-Related Disorders
11. Ophthalmological Conditions
• Squamous degeneration of the macula
12. Vascular & Circulatory Needs
• Conditions requiring rapid reorganization of blood supply
13. Animal Health & Reproductive Enhancement
• Weight gain in animals
• Increased sperm stability in storage (in animals)
14. Liver & Pancreatic Conditions
• Hepatic and pancreatic lesions
While this list seems impressive, BPC-157 would have a much easier time coming to market if one disease state was focused on and pushed through the regulatory process. Then, more off label uses could be found.
So why isn’t Diagen pushing this amazing molecule through regulation if its so effective? Are they cash strapped? It appears so, as they are actually trying to sell the BPC patent.
So then why doesn’t another big pharmaceutical company swoop in, buy the patent, and use their exhaustive funds to get BPC FDA approved, then make billions of dollars selling the drug to an already interested market. Because big pharma is evil? Not really.
If BPC-157 was purchased today, the patent would likely expire in 2033 before the phase 1-2-3 trials would be completed, at which point all the generic manufacturers would be able to push the product on the back of all the hard work a pharmaceutical company can do (I’m not sure if FDA approval would push the expiration of the patent back, but even then it would not be so financial lucrative).
On top of this, the market is already flush with BPC-157. Why would a company work so hard for this drug when people are getting it cheaply through compounded and grey/black market sources. Diagen seems unable to go after and sue all these people despite owning the patent, while Eli Lily with their deep pockets is going after GLP-1 compounders.
To complicate things further, over the last few years, several patents have come up for different use cases and applications of BPC-157. Because of the way patents for small peptides work, anyone can make a small change to a molecule, adding one amino acid here or there and now have a competing product that would not fall under the patent. For example, N-acetylated versions of BPC-157 will likely be pushed through for patent soon. Why would anyone spend a decade and hundreds of millions of dollars when tomorrow someone will make a small change to it and have their own product?
To get it FDA approved would require someone who so believes in the effects of BPC-157 to cough up a nice chunk of cash and support the research for its potential (I’m still bullish) restorative effects. To get it approved, you would need to:
1. Buy the patent (its for sale)
2. Figure out a specific disease indication to apply for BPC-157 approval
3. Hire a research team to run phase 1, 2, 3 trials
The financial incentives make it so that BPC-157 will likely never go through the full FDA approval process, which is a shame given how promising this compound is.
It is also a shame given the risk calculations in my mind for these small, short chain peptides is far different than a traditional pharmaceutical drug, given this is, in some ways, replacing/replenishing an endogenous gastric peptide that appears to decrease with age/stress. Given the human trials in early 2000’s showed no adverse events, despite using enormous doses, and the animal data corroborating this, many practitioners feel comfortable prescribing/recommending BPC-157 (I am not making such a recommendation).
The best hope for BPC is that this new regulatory environment allows for oral versions of the peptide to continue as OTC supplements (no GRAS indication has been given) while injectable forms are prescribed with modifications through compounding pharmacies as PDA or N-acetyl-BPC-157. And if we are lucky, someone with deep pockets will experience beneficial effects of this drug and decide to fund these studies themselves for the betterment of humanity.
This could easily go south if new safety concerns were brought forward for oral/injectable forms or impurities in the production by those who are seeking to profiteer off of this compound lead to adverse events. The large telehealth and compounding companies could fund this research to help people become more comfortable with using this compound.
If you’re interested, you can buy the BPC-157 patent here, before it expires early next decade. bpc157.si/bpc-157-stable…
In summary:
- BPC-157 is a promising compound for different cytoprotective uses
- It is not FDA approved for human use
- There is not some shadow cabal blocking the use of BPC-157
- Financial incentives make it so that BPC-157 is challenging to push through the regulatory process
- It will likely never receive FDA approval, but will be available in some form as oral supplement or through compounding pharmacies, with some modification
This is not medical advice. I am not suggesting you use BPC-157. It is not a FDA approved as a drug and the safety profile is still not clear. Discuss this with your doctor, with the hope that your doctor is aware of what BPC-157 is or find a doctor that is comfortable with this compound.

English