Sabitlenmiş Tweet
tya
17.7K posts


tya
@thisisangie
a PCP and polyglot 🇩🇪🇺🇸🇨🇳 - Wenn ich dir sag „ich kriege“ das hin, dann „ich kriege“ das hin
The bench and the clinic Katılım Mart 2009
185 Takip Edilen123 Takipçiler
tya retweetledi

Scientists @CellPressNews reviewed 408 YouTube clips of sleeping cats and found 2/3 curled up on their left side.
Here’s the neuroscience behind why:🧵
(Please watch their video abstract I put on the last post - it’s 100% worth it)

English
tya retweetledi

Is Aging caused by DNA damage? No.
And I’ll explain why.
In a previous post, I explained why mitochondrial damage does not cause aging. Now it's DNA damage's turn.
For decades, one of the most accepted theories held that aging was the result of accumulated damage to DNA. This idea, born after Watson and Crick’s discovery of the double helix, seemed intuitive: if DNA contains the instructions to build and maintain the organism, then the deterioration of those instructions should lead to the progressive collapse of the biological system.
In theory, it made sense. In practice, it doesn't. Today we know that this explanation is imprecise and, in many ways, outdated.
First, only 2% of the human genome is coding. The remaining 98% corresponds to non-coding regions. While there are regulatory elements in those non-coding regions (such as microRNA zones), there are also large amounts of sequences that are functionally dispensable—or even useless.
Now, if aging were caused by random DNA damage, we would expect to observe a chaotic and asymmetric phenomenon across cells and tissues, since not all cells would be damaged in the same way or at the same time. However, what we actually see is the opposite: aging is surprisingly homogeneous, synchronous, and predictable. The cells of a given tissue age in a coordinated fashion, as if obeying a common program. The odds of all cells accumulating the same damage, in the same DNA regions, at the same time, across the same tissues and species, are zero.
Chance does not orchestrate symphonies.
This brings us to a fundamental distinction: not all DNA damage carries the same biological meaning. Cancer, for example, is caused by mutations. But cancer and aging are completely different phenomena. Cancer arises from a single cell with mutations. Aging, on the other hand, is a systemic process: all cells in the body participate, and they do so in an orderly way, following specific temporal patterns that are even conserved across species.
It’s also true that if we deliberately damage an organism's DNA, we can accelerate its deterioration. But that doesn’t mean DNA damage is the natural cause of aging.
Let me explain it simply: Think of a smartphone with a built-in programmed obsolescence system. If we throw it into water, it will start malfunctioning prematurely. But not because the water accelerated the obsolescence program, or because the water is that program. We simply added entropy. In the same way, damaging DNA introduces entropy into a system that already had its own internal biological clock. It would age anyway—only now it does so under worse conditions.
Aging does not require that external entropy to occur. It’s already programmed to happen, even in organisms kept under optimal conditions.
In an aged organism, we observe DNA damage, but that damage is a consequence of aging—not its cause. The same goes for mitochondrial dysfunction, loss of proteostasis, or even epigenetic damage (we'll talk about this in a future post)—all of these are symptoms of the process, not its engine.
The key is to understand that aging is not biological entropy. It is an active, regulated, genetically programmed process. It’s not disorder.
It’s the architecture of decline.
And that detail is not minor. It gives us a strong clue about how we should act. We must intervene in the biological algorithm that activates decline. We must turn off the aging genes, restore youthful epigenetics, and reset the cellular timeline.
That's not futurism.
It's engineering.
And we're heading straight for it.
That's all.

English
tya retweetledi

This is wild!
Engineering E. coli bacteria to turn plastic waste into paracetamol (Tylenol)
@NatureChemistry
nature.com/articles/s4155…
nature.com/articles/s4155…

English
tya retweetledi

Scientists have found that tumor necrosis factor can switch macrophage #efferocytosis from immunologically silent to pro-inflammatory during #sepsis and systemic inflammation. @LabPoltorak
Learn more in Science #Immunology: scim.ag/45Yvj6Y
English
tya retweetledi

Iron deficiency causes aspartate-sensitive dysfunction in CD8+ T cells @NatureComms @MeganRTeh @Drakesmith_Lab
nature.com/articles/s4146…

English
tya retweetledi

Here's a tutorial I wrote on how I design detection and cloning primers using Primer3. Hopefully it's helpful for students. 💚
binomicalabs.notion.site/PCR-Primer-Des…
English
tya retweetledi

T follicular helper cells can play unconventional immunoregulatory roles, such as supporting cytotoxic #Tcell responses and producing effector-like cytokines to shape #Bcell fates, according to a new Review.
Read more in Science #Immunology: scim.ag/3SQqyoq
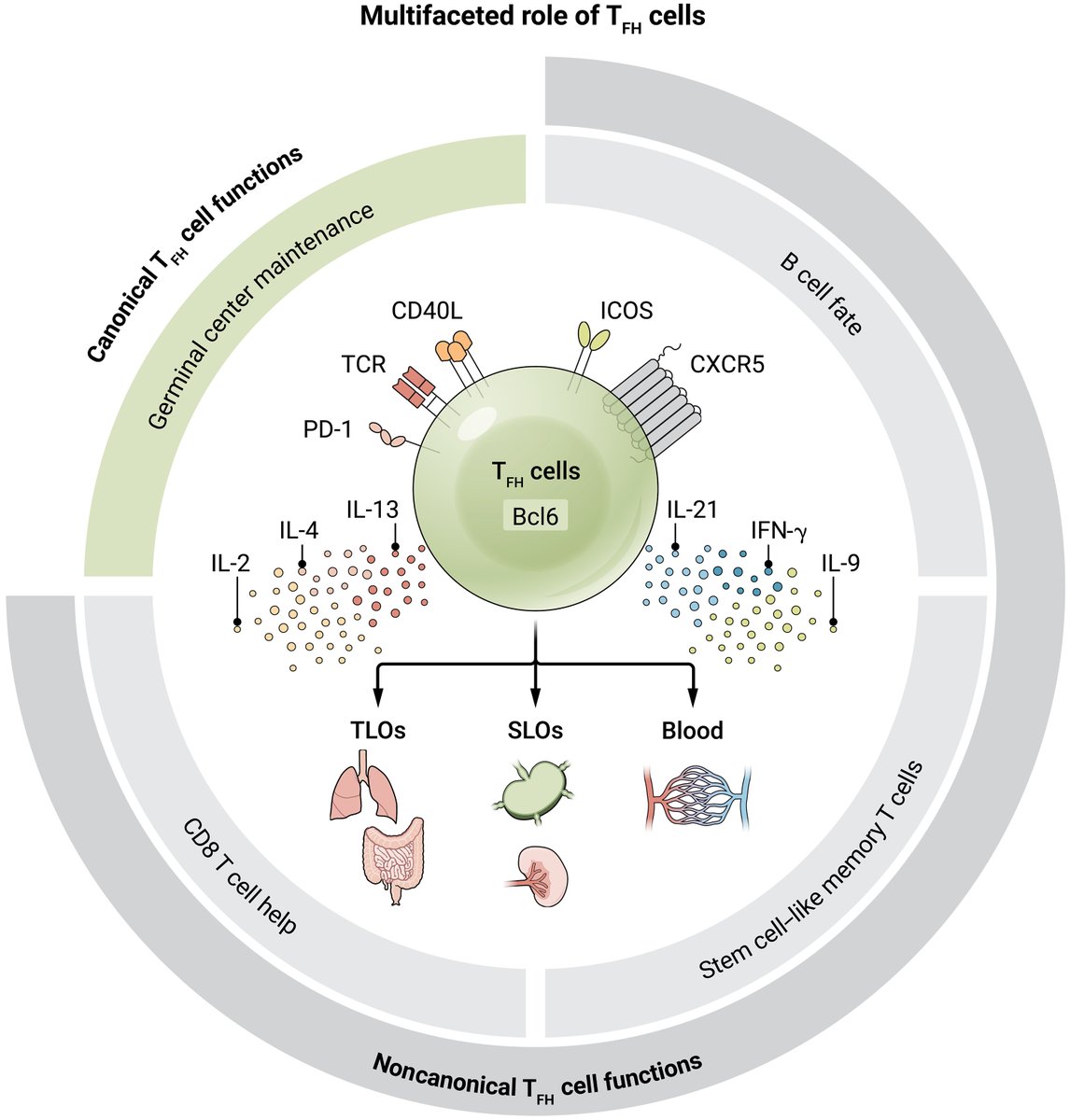
English
tya retweetledi
tya retweetledi

Cancer vaccines will be a big part of the future of immunotherapy. A new, super review @TheLancet
thelancet.com/journals/lance…
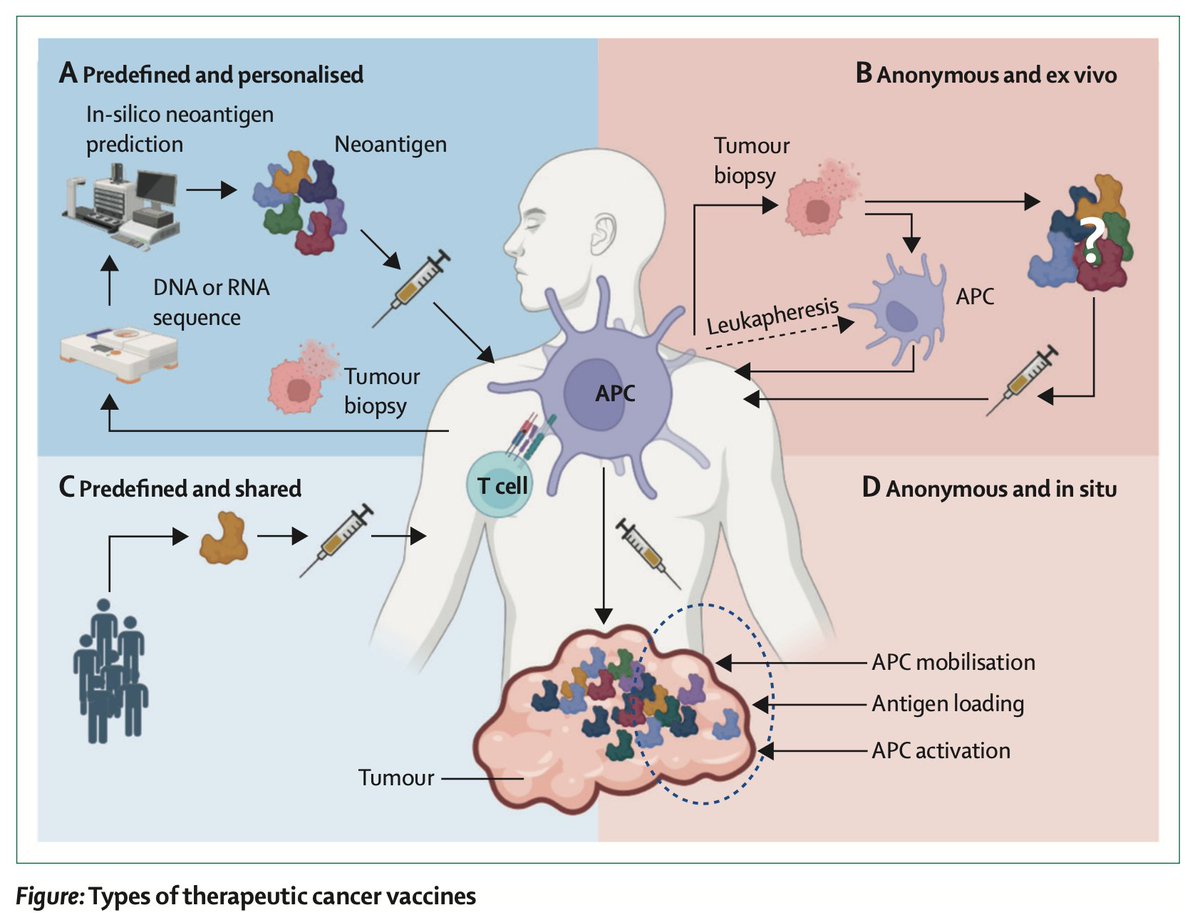
English
tya retweetledi

Maternal vitamin D regulates the metabolic rearrangement of offspring CD4+ T cells in response to intestinal inflammation @CellReports
cell.com/cell-reports/f…

English
tya retweetledi

A new Science #Immunology study suggests that cancerous cells that express PSGL-1 can escape #phagocytosis by blocking integrin signaling in #macrophages. scim.ag/3HFr1Y7

English
tya retweetledi
tya retweetledi

Lung tumor immunity: redirecting macrophages through infection-induced inflammation dlvr.it/TLK3c9
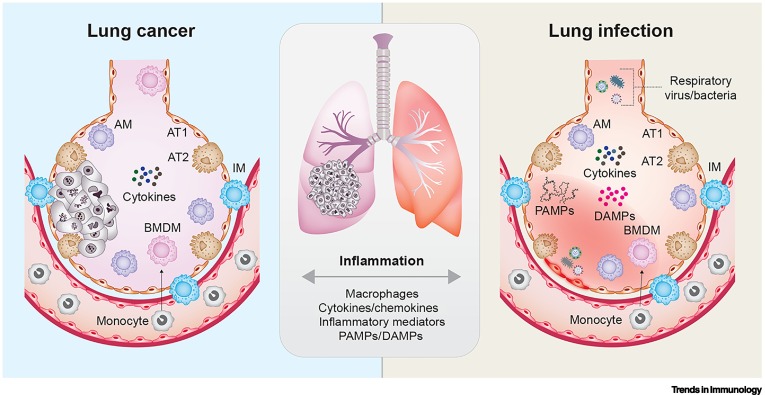
English
tya retweetledi

How exercise protects the brain through neurovascular-associated astrocytes and neuroplasticity:
Results of elegant single cell/nuceli studies in the experimental Alzheimer's model @NatureNeuro
nature.com/articles/s4159…
English
tya retweetledi

A new Science #Immunology study suggests that lipid nanoparticle #vaccines encoding the cytokine #IL12 enhance CD8 memory #Tcell function and boost immune responses in mouse models of melanoma and bacterial infection. scim.ag/43UkOiC

English
tya retweetledi
tya retweetledi

tya retweetledi

𝐈𝐦𝐦𝐮𝐧𝐨𝐠𝐞𝐧𝐢𝐜𝐢𝐭𝐲 is the ability of a substance such as an antigen or epitope to trigger an immune response in the host.
To learn more about this NEJM Illustrated Glossary term, read the Clinical Implications of Basic Research article “Novel Proteins to Neutralize Venom Toxins” from @UniversidadCR: nej.md/4kKngiv
Explore more terms: nej.md/glossary
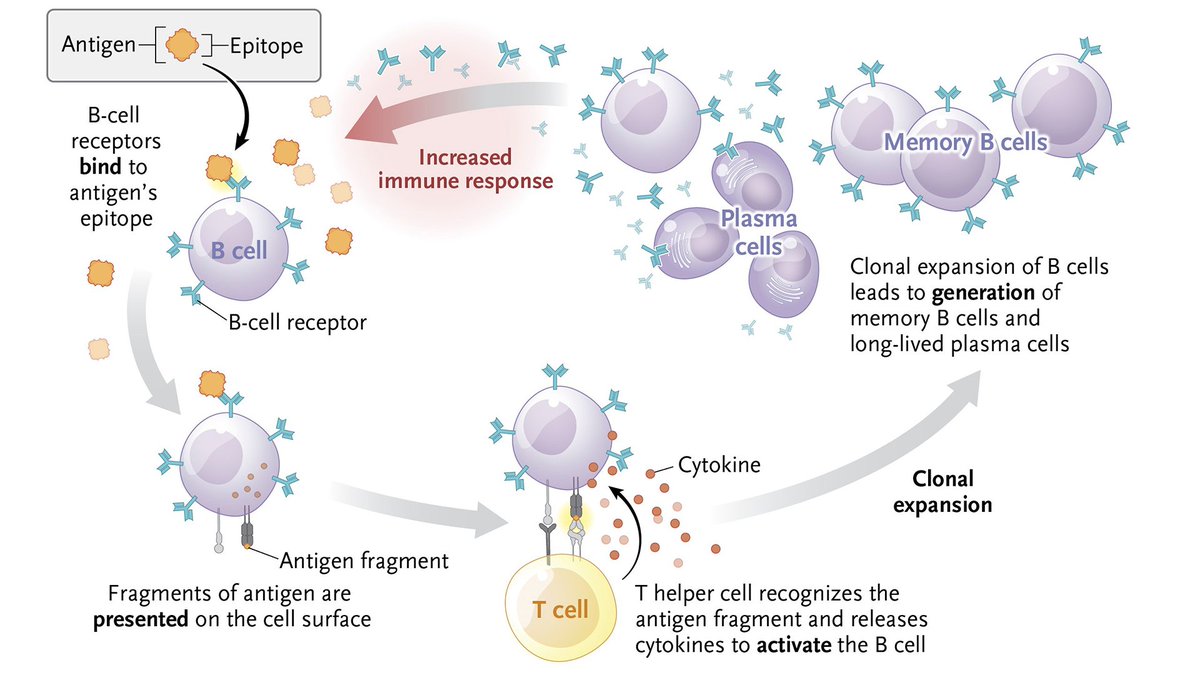
English
tya retweetledi
tya retweetledi