Patrick Erickson
98 posts


Patrick Erickson
@DrPatrickE
🐙 postdoc in the levin lab at tufts studying cell learning, aging




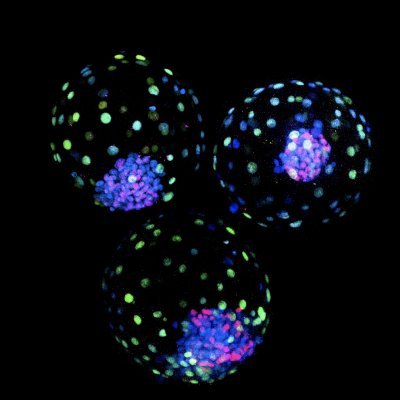
Ever wonder what a nervous system would look like if it self-assembled inside a novel being that hadn't faced a history of selection for its organism-level form and function? Or, perhaps you wondered how #Xenobots would look and act, or what their transcriptome would be like, if they had nervous systems? Well, here's the first step: advanced.onlinelibrary.wiley.com/doi/epdf/10.10… "Engineered Living Systems With Self-Organizing NeuralNetworks: From Anatomy to Behavior and Gene Expression" Our awesome team: led by @halehf: @LaurieONeill99, @mmsperry, @LPiolopez, @DrPatrickE, and Tiffany Lin. The @TuftsUniversity and @wyssinstitute press releases are here, for summaries: now.tufts.edu/2026/03/16/sci… wyss.harvard.edu/news/toward-au…








Professor Freeman Dyson on the PhD system. His talks with me are one of the reasons I did not peruse a PhD or academia directly. “The PhD system is an abomination...it has ruined many lives”—2016 His views were not well received by many of his peers.

Science does not advance by regression to the mean (tending to the average). Like evolution there is an outlier that harbors a solution to new challenges. Defend the outlier, despite the pressure of the statistics. That... over time... leads to progress.