William Cromwell, MD
341 posts

William Cromwell, MD
@Lipoprotein
Lipidologist with over 30 years of clinical experience. Cofounder @PreciseHlthRpt. My passion is creating tools that optimize individual cardiometabolic care.









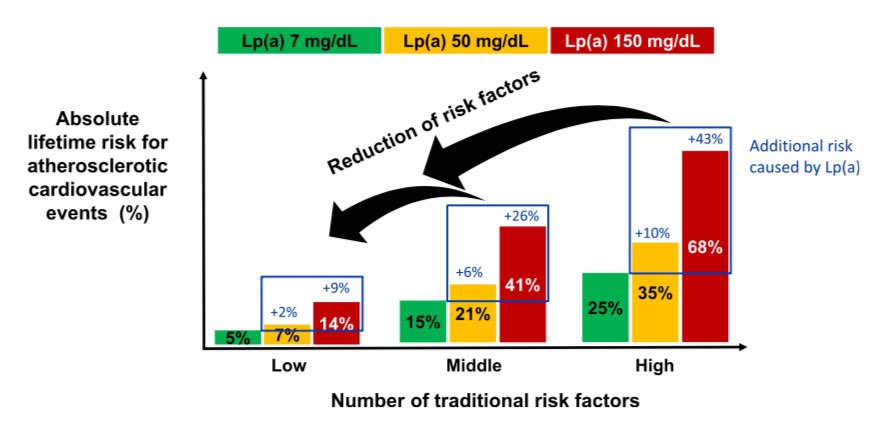






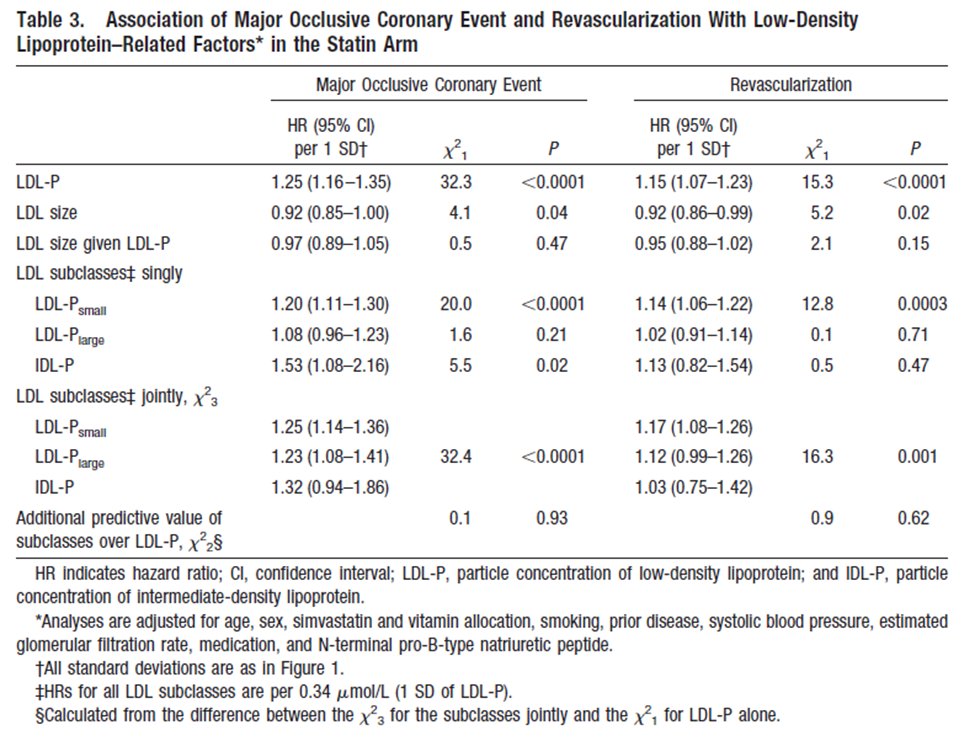











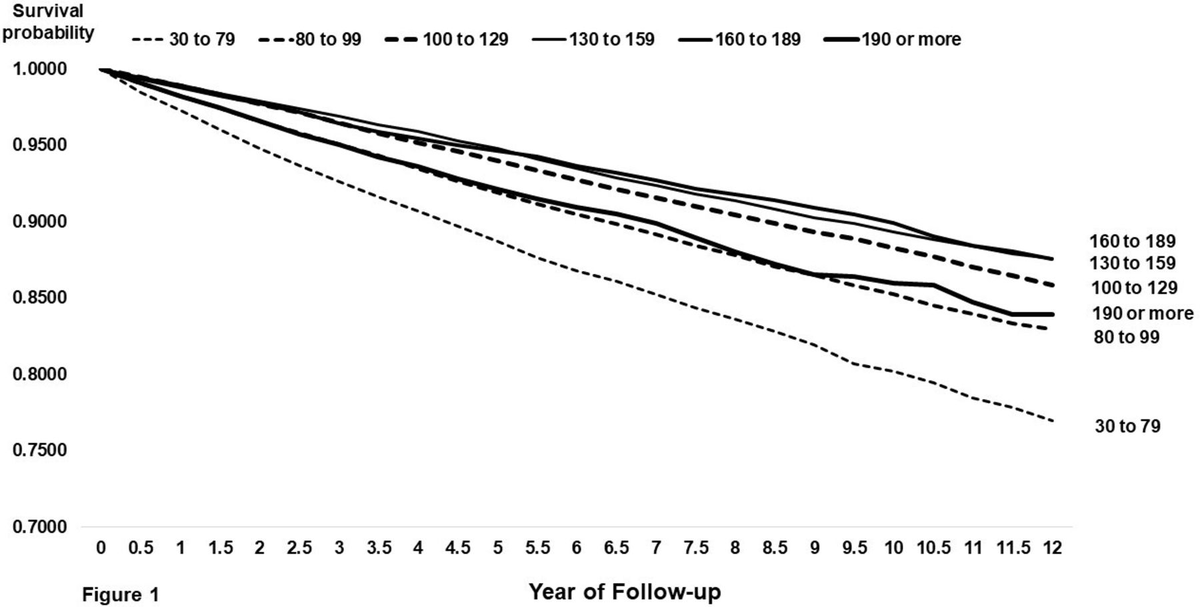
☠️ApoB and All-Cause Mortality☠️ 🚨My Most important Tweet (and video) of the Week WATCH (10m): youtu.be/sAjDjLJUlQQ High level: There is controversy over the relationship between ApoB and All-Cause Mortality (ACM), with some noting a J-curve whereby at lower levels of ApoB mortality is higher. BUT... 👉We must ask: what are/is the major driver of ACM? 👉Answer: Metabolic Vulnerability Metabolic Vulnerability can now be quantified with a multi-marker (MVX), which constitutes a metabolic "signature" suggestion a complex of nutritional and inflammatory disfunction. And MVX is GREAT at predicting ACM. One can conceptualize MVX as the background noise if one is looking at the impact of ApoB on ACM: it's like dropping a pebble into white water rapids... you won't see a ripple because the background is too strong. BUT, if you "still" the water (account for MVX), the relationship can change... and the J-curve can transform into a straight line! What are the consequences? Additional nuances? You'll have to watch the video to find out! I review: 👉Signal-to-noise ratio with respect to ACM 👉Drivers of Fatal vs Non-fatal events 👉Metabolic "Signatures" 👉Weighing the costs of intervention Major ht/ to @Lipoprotein (for references and education) and @theproof (for great 5 hour chat that you should definitely check out when it drops on his podcast... ⚠️WARNING: WILL BE PROVOCATIVE⚠️) Please engage. Watch the full video, and comment thoughtfully: youtu.be/sAjDjLJUlQQ