

Matt Kaeberlein
5K posts


@mkaeberlein
Husband, father, scientist, athlete (sort of), CEO, recovering academic. Passionate about helping people and companion animals live longer, healthier lives.






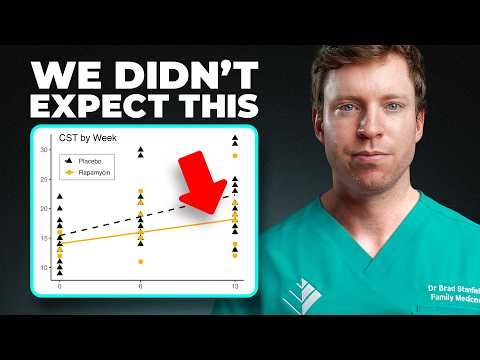








I want to start by commending @BradStanfieldMD for the tenacity required to fund and execute this trial. That’s not easy to do, and it’s a meaningful contribution to the field. The topline result, as Brad has shared, is that 13 weeks of once-weekly rapamycin did not improve muscle function in older adults and may have somewhat attenuated what many would consider “newbie gains” for untrained people beginning a dedicated training program. That said, just as we should avoid overinterpreting positive findings from a single clinical trial, we also need to be careful not to overinterpret negative ones. This is where Brad and I diverge a bit. In my view, these results represent a moderately cautionary signal—worthy of further study—but given several important limitations, they are better viewed as hypothesis-generating rather than confirmatory. A couple of alternative interpretations are worth considering: 1. Time horizon matters. My hypothesis is that the apparent attenuation in functional gains is likely a short-term effect. If the study had extended to 12 months instead of 13 weeks, I would predict the rapamycin group to show improvement. I served as an advisor for this trial, and from the outset, I advocated for a follow-up washout period of equal length, during which participants would continue their training off rapamycin. Unfortunately, funding constraints made this impossible. Importantly, the preclinical literature includes many examples where transient rapamycin exposure followed by withdrawal leads to durable improvements in age-related function—particularly in heart, ovary, oral cavity, and immune system. Mechanistically, this may reflect enhanced autophagy and reduced chronic inflammation. Given that mTOR activation is required for muscle protein synthesis, it’s not surprising that early hypertrophic responses could be blunted. But over longer timeframes—especially with intermittent or cycled dosing—it is entirely plausible (and, in my view, likely) that rapamycin could ultimately enhance functional outcomes in older adults. This is consistent with work from Ben Miller and others studying aging muscle. 2. Off-label use is more nuanced than “never.” I also disagree with the blanket position that rapamycin should never be prescribed off-label. While we absolutely need better clinical data, there is already a growing body of evidence—along with clinical experience—suggesting benefit in specific contexts. Examples include: - An ~75% responder rate reported in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS patients) - Numerous case reports of improvement in different autoimmune conditions - Early signals for improvement in age-related declines in oral health and ovarian function - Data suggesting improved cerebral blood flow and favorable brain volume changes in ApoE4 carriers - Two reasonably large phase 2 clinical trials showing improved vaccine response from everolimus For patients facing these conditions, under appropriate physician supervision and with full informed consent, I believe it is ethically appropriate to consider off-label use. Bottom line: This study adds valuable data, but it’s one piece of a much larger puzzle. The right takeaway is to refine the questions we should be asking—and to invest in the longer, better-powered trials needed to answer them.


@statto @BradStanfieldMD Interesting results but why would anybody remortgage their house for this?




Our Rapamycin & Exercise clinical trial has just been published! The topline result? Rapamycin didn't help. Instead, it may have made things worse. Here's what we found 🧵 onlinelibrary.wiley.com/doi/epdf/10.10…











