
Patrick Malone, MD PhD
1.1K posts

@patricksmalone
physician-scientist turned biotech investor @KdT_Ventures | helping founders build science and tech-driven companies | writing at https://t.co/w5M8DV8u92





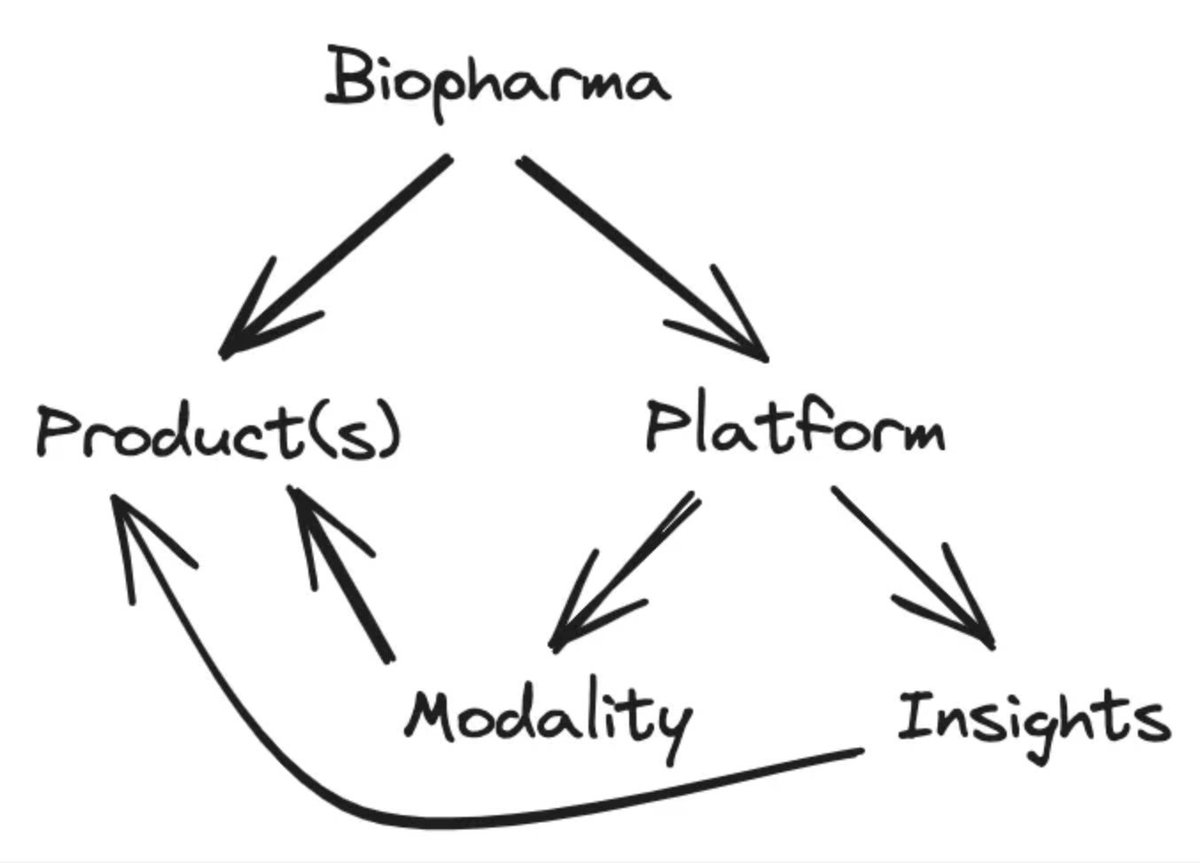
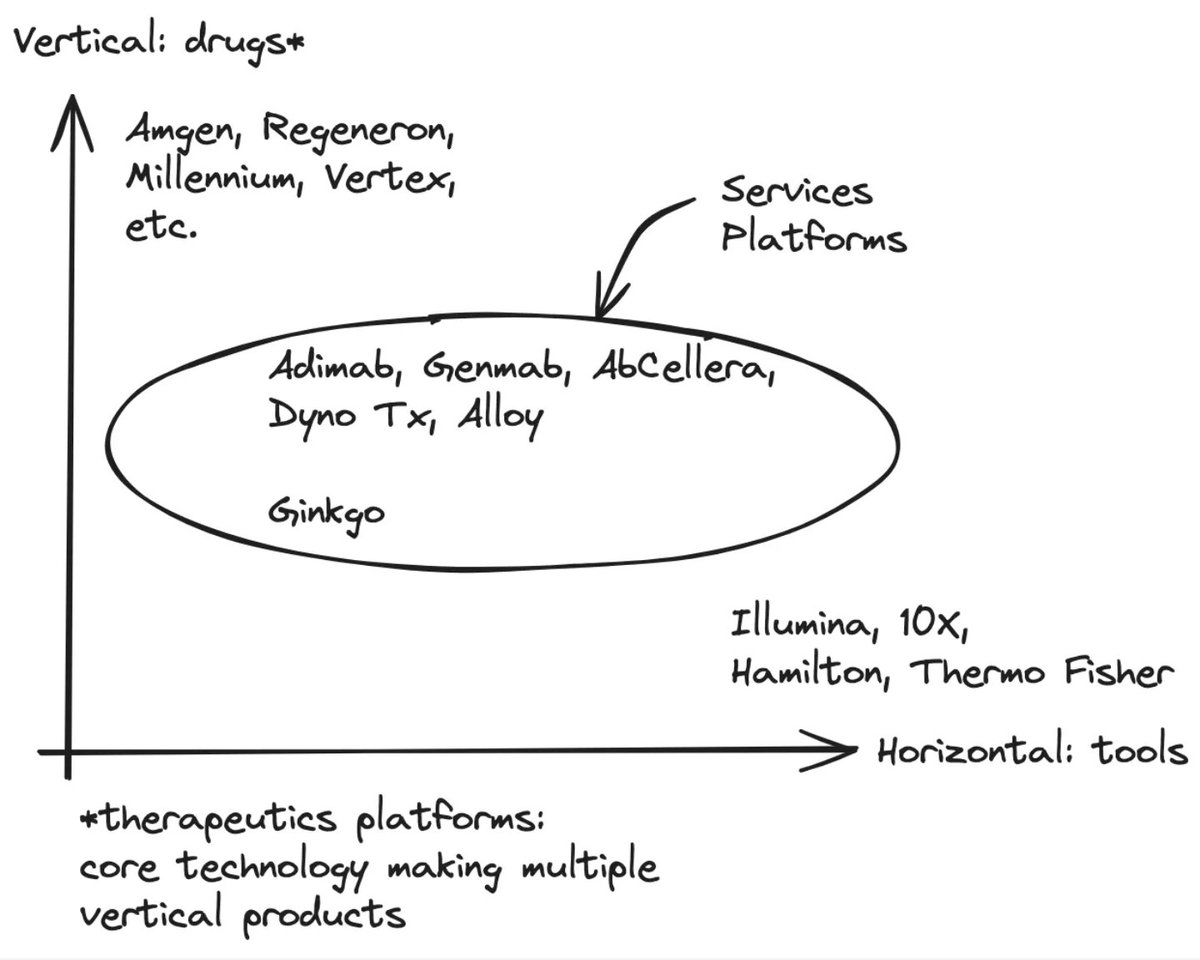
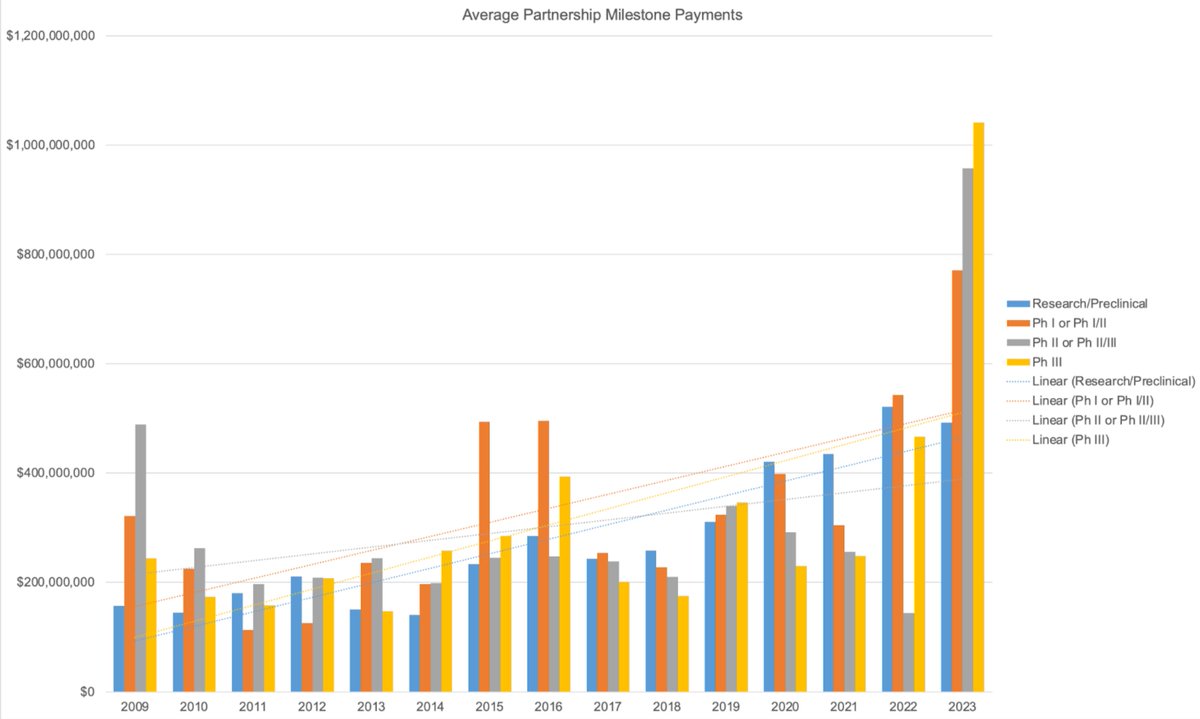
one frustrating aspect of drug development is that the incentives favor “me-too” programs that chase already-validated targets (e.g., TYK2, GLP-1, PD-(L)1). this is in large part due to the fact that patents protect molecules, not the biology behind them. once a target shows human efficacy, capital piles into copy-cat programs, while truly novel pathways and biology—often tied to the toughest diseases—go unfunded. one idea to shift the incentives: what if we broadened patent protection to include novel biology? give the first team that proves human modulation of an un-drugged pathway a time-limited (8-12 yr) marketing-exclusivity right for that target. others can still research, but they must license the “pathway patent” to launch commercially—similar to orphan-drug or priority-review vouchers. this would finalize incentive investors and drug-developers to take more target risk, and would result in faster progress and more first-in-class therapies in higher-unmet-need diseases with complex biology.

BIG NEWS: I'm thrilled to announce that Arkaea Media Group has acquired Decoding Bio, one of the leading media brands in biotechnology. This is our first acquisition and a continuation of our thesis to build media for the most consequential industries in the world. Decoding Bio was founded by @ameekapadia, @pablolubroth, @KetanYerneni, @patricksmalone, and @morgancheatham and built into the definitive voice covering the intersection of AI and biology. The opportunity in biotech media is concentrated in one place: the intersection of AI and biology. This is where the future of the industry is being built. AI is rewriting how drugs get discovered, how trials get designed, how proteins get engineered. Frontier AI labs are racing into pharma. Pharma is responding with nine and ten-figure commitments. A new generation of companies is being built by founders who grew up on both sides, and they're moving faster than the legacy industry can absorb. The media covering this shift is broken. Legacy biotech publications miss the AI story. Tech publications don't take biology seriously. The intersection between them is where critical work is happening. Decoding Bio is the only brand covering it with real credibility. To Amee, Pablo, Ketan, Patrick, and Morgan: thank you for building such an incredible brand and for choosing to build the next chapter with us. The best is ahead.





🚨 Major boost for US biotech: @WhiteHouse backed @US_FDA proposal for an **optional risk-based Expedited IND pathway** — slashing Phase 1 timelines to first-in-human trials using validated preclinical data. Reduces duplicative requirements that drive longer/higher-burden US timelines vs. China/Australia (where early trials can be 50-60% cheaper & start in weeks, vs. US delays of months to a year). Saves significant time & money for smaller firms. #Biotech #FDA


The NIH and Pharma issues are ones of leadership, not money. And the enemy is us, not China. Both the US/EU and Chinese Pharma are dependent on US academic research for discovery. Maybe US strategy needs updating, especially given the expected impact of Most Favored Nation drug pricing? It’s too early to say China is dominating Pharma. The success rate of the US/EU Pharma pipeline is only ~15% and there may be insufficient evidence that China’s is similar. Eg, if fear, quotas, and deadlines are used as strong incentives, China’s pipeline may have a significantly lower impact. If they cut corners with the ethics of human subjects, there would be bigger problems and public trust could collapse. As far as I know, the Chinese advantage is regulatory, mainly in clinical trials, but also in scientific facets. I don’t understand what the linked tweet is suggesting the US must do on “translational infrastructure”. If it’s blurring the line with patient care and drawing even more of the decades-long increasing share of the NIH budget, it seems problematic. Rather, the medical education, administration, and primary care should be done appropriately but separately from NIH funding. What about biotech? While it represents myriad models, it’s generally an intermediary and testbed between academic science and industry. If China were funding independent discovery in its biotech, it could exceed its role in the US. In the ideal case, it could supplant the need for NIH discovery and therapeutic target validation in critical areas. There is a constant assumption that NIH/academia and Pharma are not only the best there is, but that they achieve approximately the most advancement possible. I spent a lot of time in both and believe biomedical science is akin to astronomy for the 1,300 years between Ptolemy and Copernicus. A more current analogy is the visions of rockets of the US government, industry in general, and SpaceX. I regularly make the case we’re on the wrong track (as here: x.com/carlosealvare1…). One of my arguments is that NIH/academia have retreated from Enlightenment ideals for decades (as here: x.com/carlosealvare1…). The silver lining to competing with a communist superpower is that it is concerned with power rather than truth. Without freedom, including property rights, there can be no pursuit of truth. Once that is restored in US science, there’s no limit to its potential.



"Why does our FDA still incentivize all of this innovation to go to China?" @zachweinberg: "You can go to China, you can run a first-in-human study in a Chinese population at a Chinese hospital, you get your result, and then you can take that result back to America and skip the line." "I don't have to redo that Phase 1 and Phase 2 in a Western nation. I can use my Chinese data to open a Phase 3 study here and go for an approval." "Think about the incentive structure for a US biotech. You have to go to China. There is no alternative path because you've got competition on the other side who is racing ahead with infrastructure that you can't use." "We don't inspect, we don't audit, we don't send inspectors to these clinical trial sites. We have no idea what's actually going on." The Hill & Valley Forum 2026 @HillValleyForum












@patricksmalone Hard to find because most biotech CEOs want more sleep / time with family than working on policy With that being said policy is pertinent for the American edge and something we drastically need to change (aka get out of our own way) The harsh truth is that $$$ lobbying needed


Finally listening to $LLY CEO Dave Ricks (not me) with @patrickc and @collision on Cheeky Pints Literally in the first 5 minutes he echos my thoughts on what makes $RXRX a short Their entire approach is garbage in garbage out