Flamekat
1.4K posts








TWO DAYS after receiving the application, FDA greenlit the new pancreatic cancer drug daraxonrasib for expanded access. This drug has also been granted a national priority voucher, which means a full application could be reviewed in a matter of weeks instead of 10-12 months.

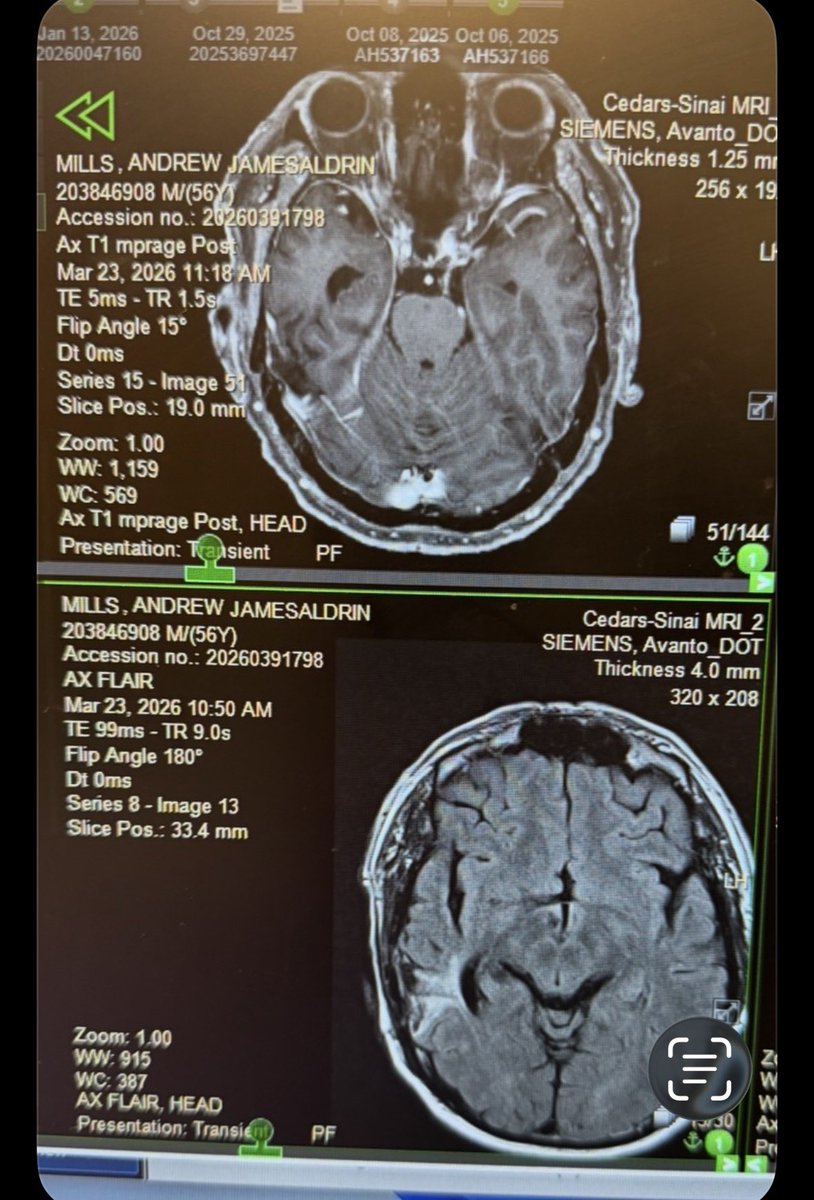
My husband was diagnosed with glioblastoma and given 12 months to live. He has been on #ANKTIVA and PDL-1 NK infusions. He has now had 2 normal brain scans. The red tape to ground breaking therapy needs to be cut! The fact the FDA wrote Dr Pat a letter to silence him is a disgrace. People deserve to choose the path that makes sense to them. They deserve hope! They shouldn’t have to fail standard therapy to be given a chance at life. The first MRI is before, the 2nd after. @bullishbruk
















