
@AbudBakri Great history of BPC-157, the world's most popular peptide!
English
Minicircle
52 posts


@MinicircleDNA
Plasmid gene therapy for health and wellness ❤️











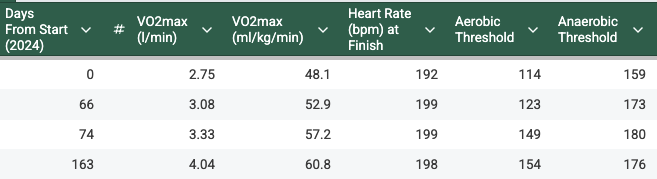









A safer approach that adds gene function without editing your genome. Watch the full episode with NFL-player-turned-scientist, Dr. Ryan Rossner, on how hypoxia and gene therapy improve longevity: youtu.be/NxRKcPa6EXw #LongevityJunky #GeneTherapy #plasmid #genome

The future of muscle growth is here, and it's more accessible than you think! Our interview with Dr. Ryan Rossner of @Minicircle dives into Follistatin via plasmid technology. A massive thanks to Dr. Rossner for sharing this groundbreaking science! youtu.be/iaTEK_GizDc

