Lauren Holder@laurencurehd
As a person with Huntington’s disease who would also be eligible for the clinical trial and has been provided the actual details from the sponsor, here is my response as to why this "sham" surgery is unethical:
Okay, so - when participating in a clinical trial with a placebo arm to it to act as a control, a pill vs a "sham" surgery is very different. Even our clinical trials where the drug is delivered via spinal tap is different because they aren't gene therapies; they are gene-modifying drugs that leave your system and you can have a "washout period", which means the drug is completely gone. The benefit of this (especially in rare diseases) is that people are able to participate in future research since the drug can actually "washout". It's still considered unethical in HD, though, to expect that someone receive a placebo (sugar pill instead of actual drug) and be "blinded" (not knowing if you received the real thing or not) for too long, because a lot of times, these treatments work better the earlier you are in the disease. If you "blind" people for too long, they then may not be eligible not only for the real drug in an open-label extension, but they could also be excluded from other clinical trials because they've progressed too much to meet the clinical trial criteria, which is very strict.
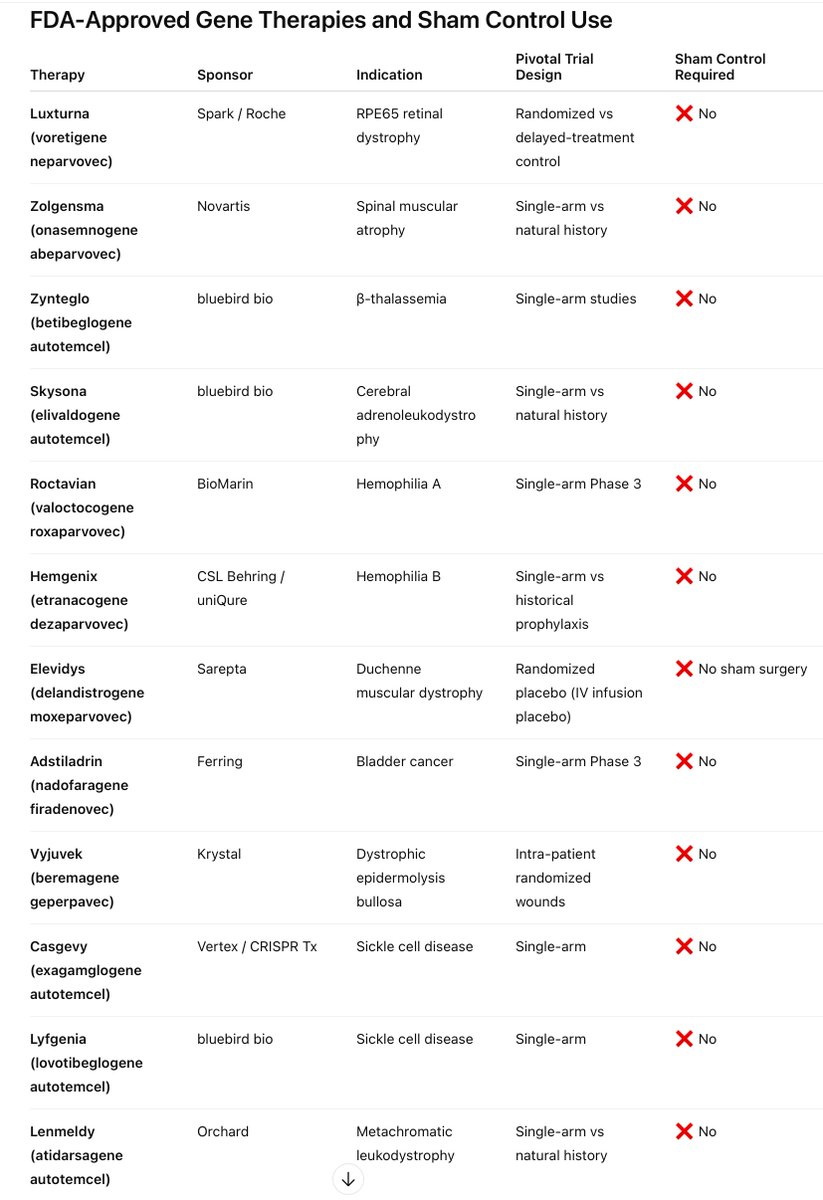
So the issues with the "sham surgery" placebo arm are:
1- This is a one-and-done gene therapy. There are not multiple treatments. The first people in the AMT-130 trial have been followed for 4+ YEARS and counting. It doesn't wash out ever. Because of that, people who participate in this clinical trial will never be able to participate in future research. Imagine doing that to someone and making them have a "sham" surgery, disqualifying them from future research and not giving them a chance for a treatment at all.
2 - why couldn't they just do the real surgery after the sham and give them the real thing? Great question! That was actually tried, but they found that the people who did sham progressed during the trial to a point they were no longer eligible for it. So very few were able to actually do that, which is the one of the reasons the FDA agreed in December 2024 that UniQure could instead use natural history data instead of doing the "sham" procedure. It's unethical in a rare, fatal neurodegenerative disease. This decision WAS ALREADY MADE BY THE FDA in 2024.
3 - The real surgery to receive the real drug involves going under anesthesia for up to 15 hours, having burr holes drilled (very common, standard procedure already done every day for other things, but still a hole in the head that requires anesthesia and a surgical room), delivering the drug right to the part of the brain affected by HD that this drug targets. After surgery, the participant then has to recover in the hospital and be monitored for several days inpatient before they are allowed to go home. They will follow up regularly for the next 5+ years to see how they are doing, if they are progressing, how much they are progressing, etc. Now - imagine going through ALL of that still - the only difference is that instead of delivering the drug into your brain, they stop after making the incisions where the burr holes would be, let you stay under anesthesia for the same amount of time and everything, put sutures at the incisions and that's it. They don't tell you whether you received the treatment. You still have to go through recovery and monitoring just like the real thing. It is absolutely evil to do that to someone with HD. It has already been proven and accepted that they will progress faster because the data shows after almost 4 years of monitoring that it slows progression of the disease by 75%. So to ask NOW, at this point, when we already know this, to ask people with HD to undergo a "sham" surgery is not just unethical, it's evil. It's disgusting.