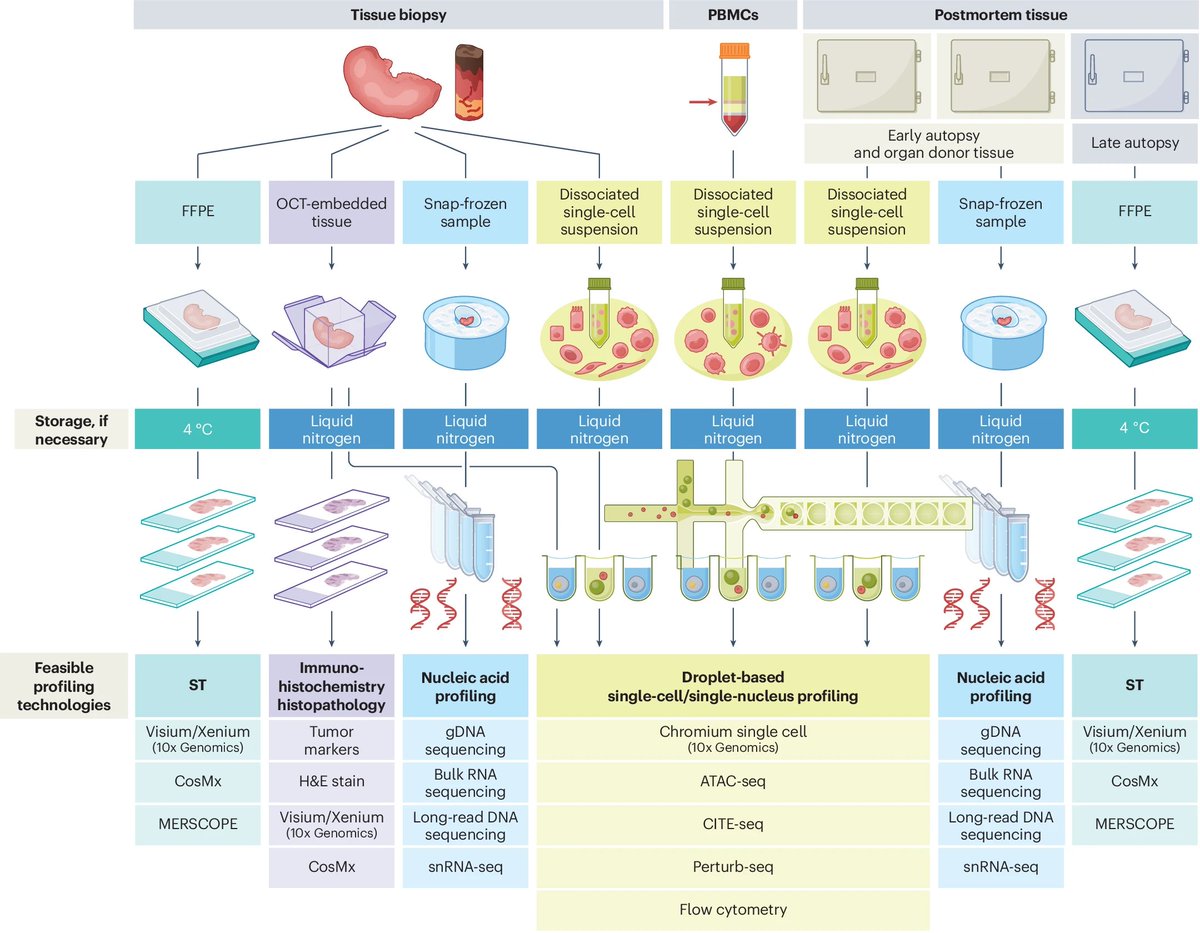
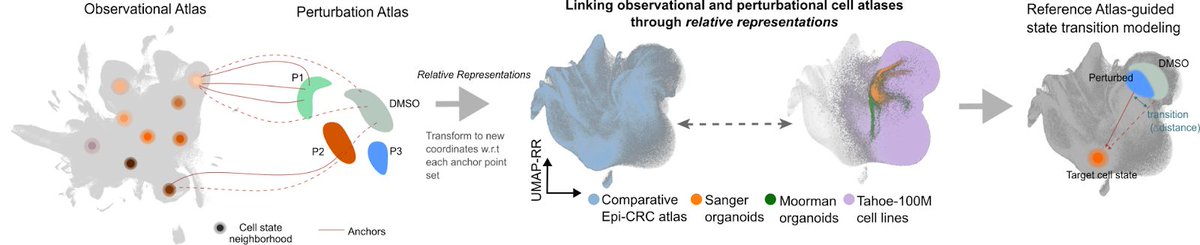
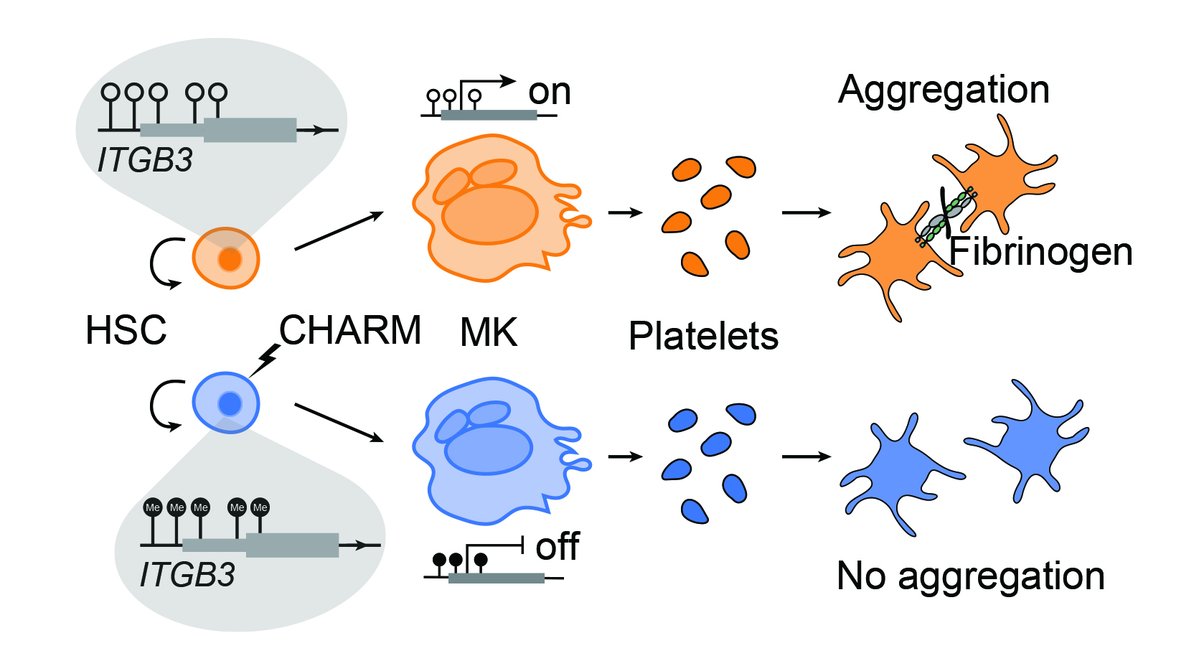
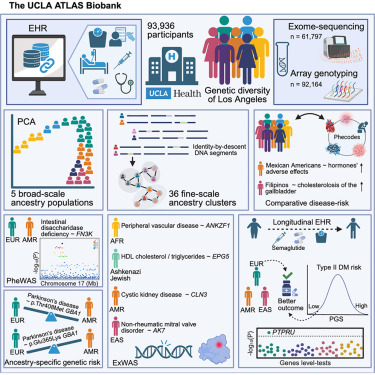
Naveen Jain รีทวีตแล้ว

We’re unwrapping a new method today @MSKCancerCenter!
Led by @SydneyBlattman, @NabihMaslah, @AustinAVarela, Ronan Chaligne and @dana_peer, plus colleagues at @10xGenomics, we’re delighted to share Genotyping In Fixed Transcriptomes (GIFT):
biorxiv.org/content/10.648…
1/n
English