
Jordan Alexander
16.9K posts


Jordan Alexander
@JD4for4
Finding hidden value in under-followed companies. Posts are not advice. Do your own due diligence.









FDA commish @DrMakaryFDA has been using exact language of @johnarnold as can be seen in this thread during recent $srpt Elevidys turmoil. When viewed through lens of Arnold being the moneyed interest behind Makary, Prasad and new hires, everything makes sense. Makary and Prasad have been mere low-level henchmen.


Podcast #378 is up! @A_May_MD on investing in biotech, $NKTR, and $ABVX yetanothervalueblog.com/p/adam-may-on-…

AV is everywhere. All VP, MM and their cousins and brothers!

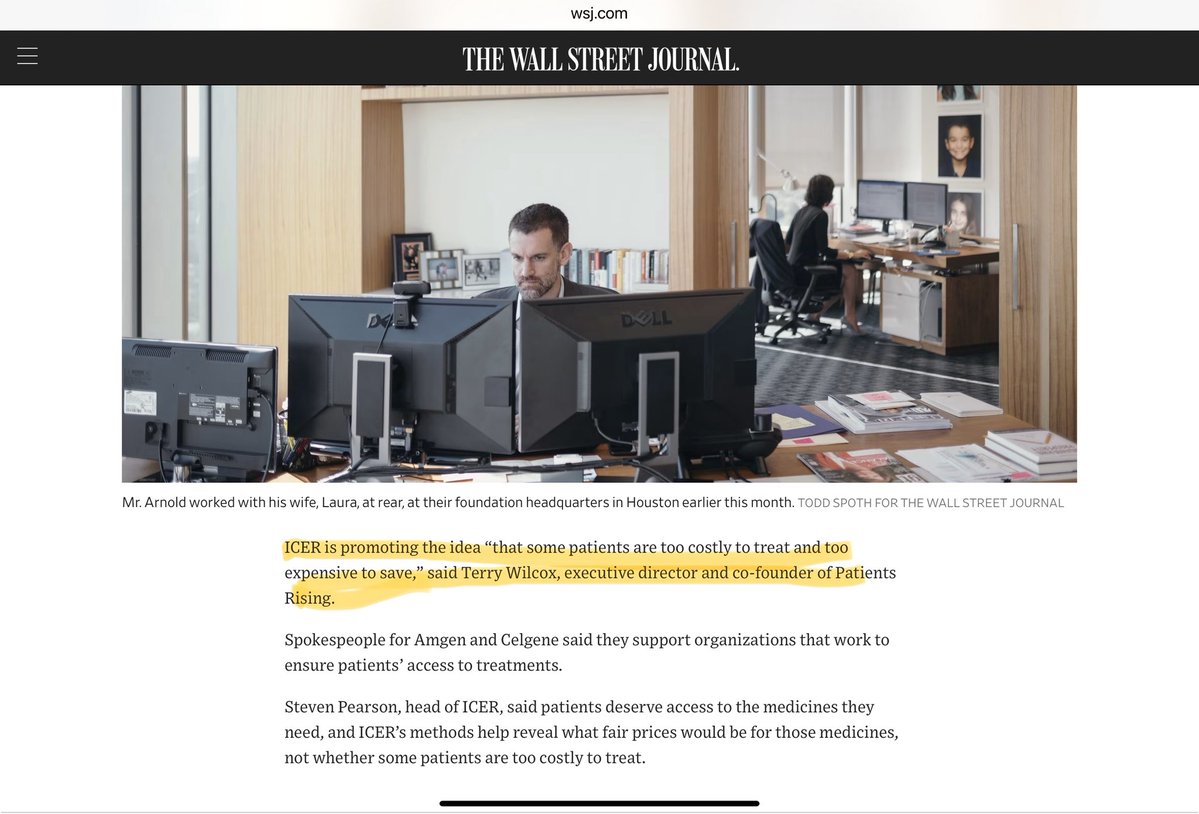
A Billionaire Pledges to Fight High Drug Prices, and the Industry Is Rattled - WSJ. Original article not generated by AI wsj.com/articles/a-bil…

This is HUGE! Vinay Prasad admitted his papers had input from John Arnold and AV. How could this bought out AV guy became CBER head? The original WSJ article is long, if there is enough interest, I can give a few nuggets.







This one prepared by Katherine Szarama at Arnold Ventures, also likely negatively impacted cell & gene therapy approvals by the current FDA before she became deputy director CBER.


These results in mice suggest that HTT1a reduction may be necessary for benefit in HD. Calls into question Skyhawk's approach, which does not address HTT1a (and supports UniQure's). science.org/doi/10.1126/sc…






A Billionaire Pledges to Fight High Drug Prices, and the Industry Is Rattled. $QURE @uniQure_NV @SenRonJohnson wsj.com/articles/a-bil…

This is HUGE! Vinay Prasad admitted his papers had input from John Arnold and AV. How could this bought out AV guy became CBER head? The original WSJ article is long, if there is enough interest, I can give a few nuggets.
