Sabitlenmiş Tweet

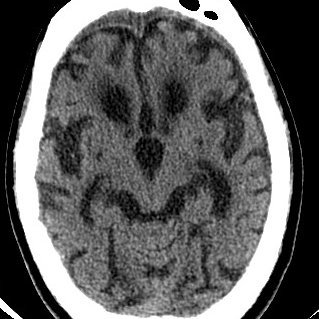
. #ICYMI doesnt this almost cure huntingtons disease? #askingforafriend #HDAwareness #CureHD #Huntingtonsdisease
LogicalValue@LogicalValueMx
$qure Tabrizi explains how they use real world data to compare AMT-130 to external control group (EnrollHD) and why regulators approve of if when done correctly.
English