R
36.2K posts
R
@Lucy3370
Looking to find hidden value in biotech's.





🇺🇸 🇨🇳 I was on @foxnewsnight tonight discussing something that should concern every American in the context of the U.S.-China summit. For decades, the United States has led the world in biotechnology. Cell & gene therapies & advanced biologics — the medicines of the future — are finally delivering real hope to patients with diseases that were once untreatable or incurable. These are not incremental improvements. They are transformative. But here’s the reality we can no longer ignore: roughly half of the global investment capital in advanced biotechnology is now flowing to China. This isn’t just about money or market share. It’s about who will control the next generation of life-saving treatments. When capital, talent, & manufacturing shift overseas, American patients lose access, American innovation slows, and our strategic position in one of the most important industries of the 21st century erodes. China is not playing by the same rules. They are moving aggressively to dominate cell and gene therapy, while we continue to tie ourselves in regulatory knots and send mixed signals to the companies trying to develop these therapies here at home. We cannot afford to watch this happen. The patients who are waiting — children with rare genetic diseases, adults facing terminal conditions, families desperate for options — don’t care about bureaucracy or politics. They care about whether these treatments reach them in time. The FDA has an essential role to play in this moment. We must modernize our approach so that we can accelerate safe and effective innovation here in America, while maintaining the rigorous standards that have made our system the global gold standard. If we fail to do this, we will not only lose our leadership — we will fail the very patients we are supposed to serve. In my career, I’ve seen what’s possible when regulation keeps pace with science, and I’ve seen what happens when it doesn’t. The video of my appearance is below 👇 . I’d love to hear your thoughts.


On the FDA, it had started to come out over the last few weeks that several political appointees that were in new leadership positions at FDA had a history of funding from Arnold family entities incl Makary. And Q's were raised re: recent drug rejections & the overruling of scientific staff which wasn't a regular occurrence at FDA until this group of political appointees came in. Some concerns were based on the history of how Arnold entities & affiliates like iCER appear to discriminate against a number of patient populations incl rare disease patients

.@drJoshS weighs in on how Marty Makary “misunderstood” the FDA. statnews.com/2026/05/16/mar…


$CLPT $QURE Karim Mikhail is the new CBER! After taking a deeper dive I think this is great News! hoganlovells.com/en/publication… Karim Mikhail’s comments at JPM were actually very interesting for rare disease and gene therapy back in Jan. He basically hinted that FDA is becoming more flexible and “not thinking in black and white” anymore. He talked about: • single pivotal study pathways • flexibility for rare disease/cell & gene therapy • accelerating approvals • reducing regulatory friction Obviously he never mentioned $QURE specifically, but if you read between the lines it’s hard not to think about Huntington disease and AMT-130. You can’t apply traditional large trial logic to terminal rare diseases with tiny patient populations and years-long progression timelines. Feels like he realized there has to be some middle ground between safety and letting people die while waiting 10+ years.



🚨Friday night shakeup at FDA: * Acting CDER chief Tracy Beth Høeg fired, replaced by Michael Davis * Acting CBER chief Katherine Szarama out, replaced by Karim Mikhael * COS Trafficant out, others too Full story here from me and @maxonwifi: endpoints.news/cder-chief-hoe…



NEW: Acting director of the drug center, Tracy Beth Høeg, is leaving the FDA, just days after Commissioner Marty Makary resigned from the agency statnews.com/2026/05/15/fda…


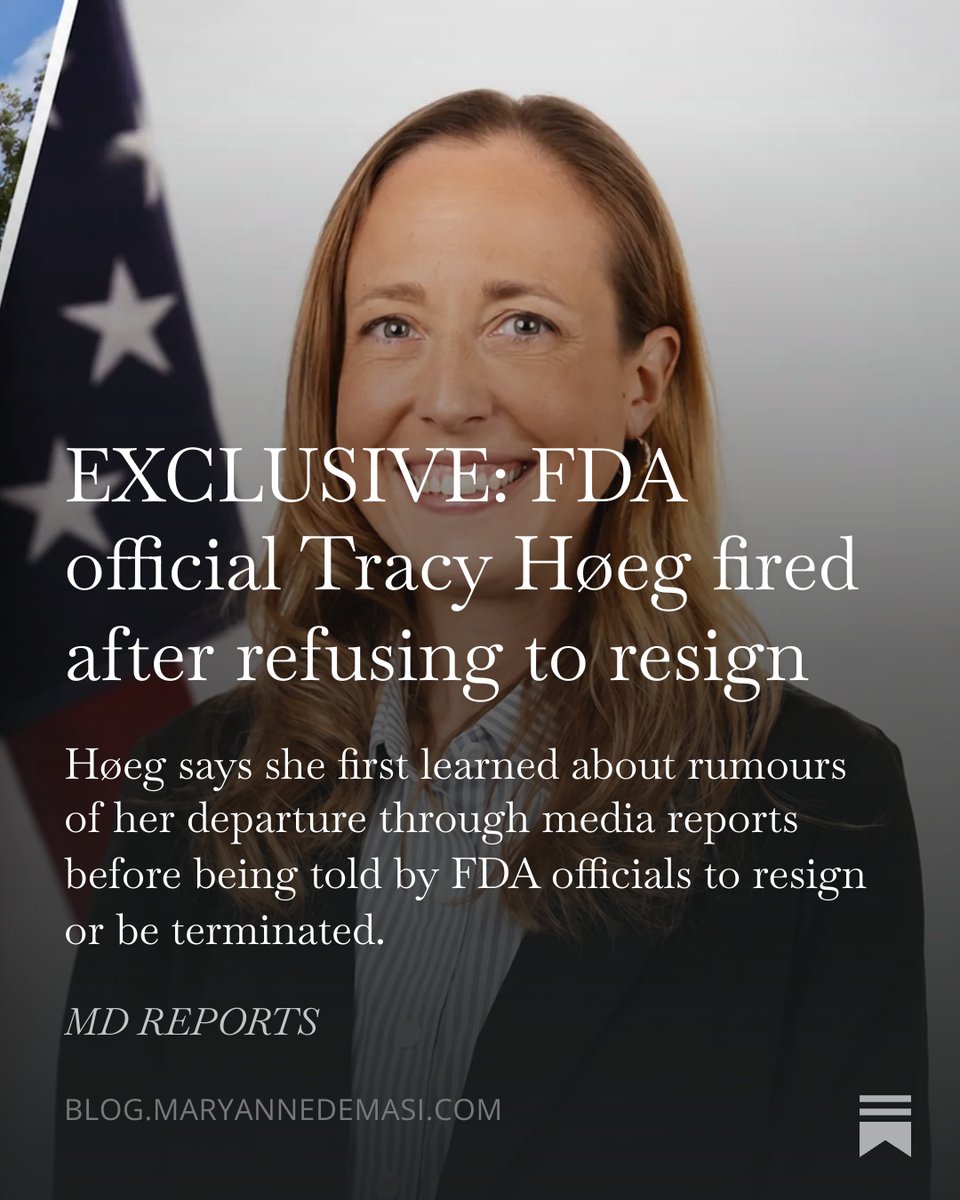

Looks like the @US_FDA is purging the Arnold Ventures taint. 👇👇👇👇 $srpt $qure $srpt